iScience ( IF 4.6 ) Pub Date : 2020-09-20 , DOI: 10.1016/j.isci.2020.101580 Weilei Hu 1 , Guosheng Wang 2 , Yian Wang 1 , Matthew J Riese 3, 4 , Ming You 1
|
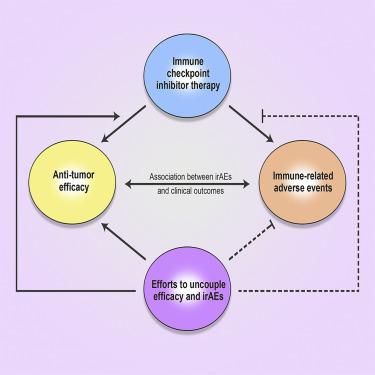
Immunotherapy with monoclonal antibodies targeting immune checkpoint molecules, including programmed death-1 (PD-1), PD ligand-1 (PD-L1), and cytotoxic T-lymphocyte-associated antigen (CTLA)-4, has become prominent in the treatment of many types of cancer. However, a significant number of patients treated with immune checkpoint inhibitors (ICI) develop immune-related adverse events (irAEs). irAEs can affect any organ system, and while most are clinically manageable, irAEs can result in mortality or long-term morbidity. Factors that can predict irAEs remain elusive. Understanding the etiology of ICI-induced irAEs and ways to limit these adverse events are needed. In this review, we provide basic science and clinical insights on the mechanisms responsible for ICI efficacy and ICI-induced irAEs. We further provide insights into approaches that may uncouple irAEs from the ability of ICIs to kill tumor cells.
中文翻译:

将免疫检查点阻断中的治疗效果与免疫相关不良事件分开
使用针对免疫检查点分子的单克隆抗体进行免疫治疗,包括程序性死亡-1 (PD-1)、PD配体-1 (PD-L1) 和细胞毒性 T 淋巴细胞相关抗原 (CTLA)-4,已成为治疗中的重点许多类型的癌症。然而,大量接受免疫检查点抑制剂(ICI)治疗的患者出现了免疫相关不良事件(irAE)。 irAE 可以影响任何器官系统,虽然大多数在临床上是可控的,但 irAE 可能导致死亡或长期发病。能够预测 irAE 的因素仍然难以捉摸。需要了解 ICI 诱导的 irAE 的病因以及限制这些不良事件的方法。在这篇综述中,我们提供了有关 ICI 疗效和 ICI 诱导的 irAE 机制的基础科学和临床见解。我们进一步提供了一些方法的见解,可以将 irAE 与 ICI 杀死肿瘤细胞的能力分开。











































 京公网安备 11010802027423号
京公网安备 11010802027423号