Chemical Physics Letters ( IF 2.8 ) Pub Date : 2020-09-16 , DOI: 10.1016/j.cplett.2020.138001 G.V. Karpov , E.S. Vasiliev , N.D. Volkov , I.I. Morozov , S.V. Savilov
|
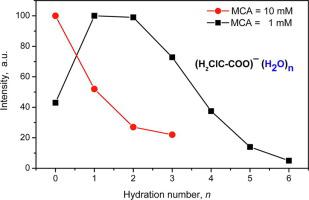
The structure of hydrated ions of monochloroacetic acid was determined by electrospray of aqueous solution of monochloroacetic acid into the vacuum chamber of the mass spectrograph. It was found that the distribution of ions in degree of hydration depends on the concentration of acid in the initial solution. The number of the hydrated ions monotonically decreases with growth of their hydration shell at concentration of the acid of 10 mM. At acid concentrations <1 mM, the distribution is peaked on the hydration number n = 2. This maximum separates water molecules with a strong and weak bonding with the ion.
中文翻译:

质谱数据分析水中一氯乙酸阴离子的结构
通过将一氯乙酸水溶液电喷雾到质谱仪的真空室中来确定一氯乙酸的水合离子的结构。已发现离子在水合度中的分布取决于初始溶液中酸的浓度。在10 mM的酸浓度下,水合离子的数目随其水合壳的生长而单调减少。在酸浓度<1 mM时,该分布在水合数n = 2处达到峰值。该最大值分离出与离子具有强键和弱键的水分子。











































 京公网安备 11010802027423号
京公网安备 11010802027423号