当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Gold-Catalyzed Addition-N-Boc Cleavage-Cyclization of N,O-Acetal with Ynamides for Construction of 6-(tert-Butyldimethylsilyl)oxy-tetrahydropyrrolo[1,2-c][1,3]oxazin-1-ones.
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-09-14 , DOI: 10.1021/acs.joc.0c01776 Yan-Xue Zhang 1, 2 , Ling-Yan Chen 1, 3 , Jian-Ting Sun 1, 2 , Chang-Mei Si 2 , Bang-Guo Wei 2
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-09-14 , DOI: 10.1021/acs.joc.0c01776 Yan-Xue Zhang 1, 2 , Ling-Yan Chen 1, 3 , Jian-Ting Sun 1, 2 , Chang-Mei Si 2 , Bang-Guo Wei 2
Affiliation
|
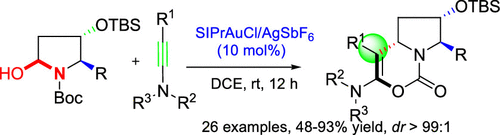
A highly efficient gold-catalyzed approach between N,O-acetals and ynamides for the construction of 6-(tert-butyldimethylsilyl)oxy-tetrahydropyrrolo[1,2-c][1,3]oxazin-1-ones was developed. This reaction tolerated a wide range of substituted N,O-acetals and TsN-substituted ynamides, leading to novel heterocycles in moderate to good yields and with excellent diastereoselectivities (dr > 99:1).
中文翻译:

金催化的N,O-缩醛与酰胺的加成-N-Boc裂解-环化反应,生成6-(叔丁基二甲基甲硅烷基)氧基-四氢吡咯并[1,2-c] [1,3]恶嗪-1-酮。
提出了一种在N,O-乙缩醛和乙酰胺之间的高效金催化方法,用于构建6-(叔丁基二甲基甲硅烷基)氧基-四氢吡咯并[1,2- c ] [1,3]恶嗪-1-酮。该反应可耐受各种取代的N,O-乙缩醛和TsN取代的乙酰胺,从而以中等至良好的收率和极好的非对映选择性(dr> 99:1)产生新型杂环。
更新日期:2020-10-02
中文翻译:

金催化的N,O-缩醛与酰胺的加成-N-Boc裂解-环化反应,生成6-(叔丁基二甲基甲硅烷基)氧基-四氢吡咯并[1,2-c] [1,3]恶嗪-1-酮。
提出了一种在N,O-乙缩醛和乙酰胺之间的高效金催化方法,用于构建6-(叔丁基二甲基甲硅烷基)氧基-四氢吡咯并[1,2- c ] [1,3]恶嗪-1-酮。该反应可耐受各种取代的N,O-乙缩醛和TsN取代的乙酰胺,从而以中等至良好的收率和极好的非对映选择性(dr> 99:1)产生新型杂环。











































 京公网安备 11010802027423号
京公网安备 11010802027423号