Chemical Physics Letters ( IF 2.8 ) Pub Date : 2020-09-11 , DOI: 10.1016/j.cplett.2020.137992 Zahoor H. Farooqi , Anam Masaud , Robina Begum , Ahmad Irfan
|
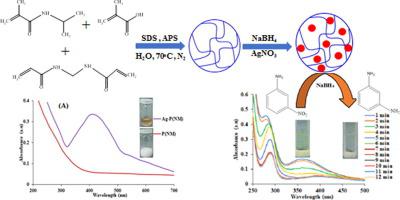
Poly(N-isopropylmethacrylamide-co-methacrylic acid) [P(NM)] network with colloidal size was obtained by emulsion polymerization. Chemical reduction of silver nitrate via NaBH4 produced silver nanoparticles within network. Both P(NM) and Ag-P(NM) were characterized. 3-nitroaniline (3-NA) was reduced with NaBH4 in presence of Ag-P(NM) to test its catalytic activity. Catalytic efficacy of Ag-P(NM) was investigated under various conditions of NaBH4, 3-NA and Ag-P(NM). Dependence of rate constant upon NaBH4 and 3-NA describes that 3-NA reduction takes place by following Langmuir Hinshelwood mechanism. Acquired kinetic data verifies that Ag-P(NM) is an efficient catalyst for reduction of 3-NA. Catalytic activity of Ag-P(NM) was found tunable via lead nitrate concentration.
中文翻译:

使用甲基丙烯酰胺基纳米杂化催化剂还原3-硝基苯胺的理化方面
通过乳液聚合获得具有胶体尺寸的聚(N-异丙基甲基丙烯酰胺-共-甲基丙烯酸)[P(NM)]网络。通过NaBH 4化学还原硝酸银可在网络内生成银纳米颗粒。表征了P(NM)和Ag-P(NM)。在Ag-P(NM)存在下,用NaBH 4还原3-硝基苯胺(3-NA )以测试其催化活性。的Ag-P(NM)的催化效力的NaBH的各种条件下研究了4,3-NA和Ag-P(NM)。速率常数对NaBH 4的依赖性和3-NA描述了通过遵循Langmuir Hinshelwood机理发生3-NA减少。所获得的动力学数据证实,Ag-P(NM)是还原3-NA的有效催化剂。发现Ag-P(NM)的催化活性可通过硝酸铅浓度调节。











































 京公网安备 11010802027423号
京公网安备 11010802027423号