Bioorganic Chemistry ( IF 4.5 ) Pub Date : 2020-09-08 , DOI: 10.1016/j.bioorg.2020.104270 Siwar Ghannay 1 , Mejdi Snoussi 2 , Sabri Messaoudi 3 , Adel Kadri 4 , Kaïss Aouadi 1
|
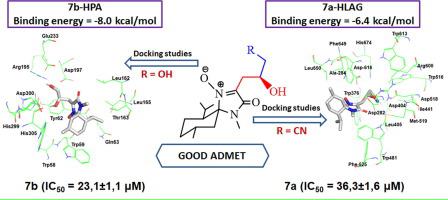
In an effort to explore a new class of antidiabetic inhibitors, a new series of isoxazolidine and C-alkyl imine oxide derivatives scaffolds were designed, synthesized and fully characterized. The newly synthesized analogues were evaluated for their human pancreatic α-amylase (HPA) and human lysosomal acid-α-glucosidase (HLAG) inhibitory activities and have shown a higher potency than acarbose. The compounds 7b (23.1 ± 1.1 μM) and 7a (36.3 ± 1.6 μM) were identified as the potent HPA and HLAG inhibitors with inhibitory effect up to 9 and 21-fold higher than acarbose, respectively. Antihyperglycemic activity results were supported by molecular docking approach of the most potent compounds 7b and 7a showing stronger interactions with the active site of HPA and HLAG as well as by in silico absorption, distribution, metabolism, excretion and toxicity (ADMET) profile suggesting their satisfactory oral druglikeness without toxic effect. Therefore, it can be concluded that both 7b and 7a can be used as effective lead molecules for the development of HPA and HLAG inhibitors for the management of T2DM.
中文翻译:

新型对映体纯的异恶唑烷和C-烷基亚胺氧化物衍生物作为潜在的降糖药:设计,合成,α-淀粉酶和α-葡萄糖苷酶的双重抑制剂,ADMET和分子对接研究。
为了探索新型的抗糖尿病抑制剂,设计,合成并充分表征了一系列新的异恶唑烷和C-烷基亚胺氧化物衍生物支架。评价了新合成的类似物的人胰腺α-淀粉酶(HPA)和人溶酶体酸-α-葡萄糖苷酶(HLAG)抑制活性,并显示出比阿卡波糖更高的效力。化合物7b( 23.1± 1.1μM )和7a( 36.3± 1.6μM )被鉴定为有效的HPA和HLAG抑制剂,抑制作用最高可达9种。比阿卡波糖高21倍。最有效的化合物7b和7a的分子对接方法显示出与HPA和HLAG的活性位点之间更强的相互作用,以及计算机吸收,分布,代谢,排泄和毒性(ADMET),表明它们具有令人满意的降血糖活性口服药物似无毒性作用。因此,可以得出结论,7b和7a均可用作开发HPA和HLAG抑制剂以治疗T2DM的有效先导分子。











































 京公网安备 11010802027423号
京公网安备 11010802027423号