当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Enantioselective addition of thiols to trifluoromethyl ketimines: synthesis of N,S-ketals.
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-09-08 , DOI: 10.1039/d0ob01725f Xiaonan Wang 1 , Yuan Gao 1 , Zhonglin Wei 1 , Jungang Cao 1 , Dapeng Liang 1 , Yingjie Lin 1 , Haifeng Duan 1
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-09-08 , DOI: 10.1039/d0ob01725f Xiaonan Wang 1 , Yuan Gao 1 , Zhonglin Wei 1 , Jungang Cao 1 , Dapeng Liang 1 , Yingjie Lin 1 , Haifeng Duan 1
Affiliation
|
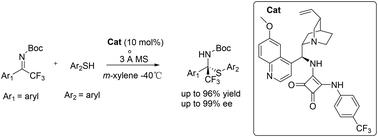
An efficient enantioselective addition of thiols to acyclic trifluoromethyl ketimines has been established by using a bifunctional squaramide catalyst, which was derived from quinine, and the reaction was completed in 5 to 10 min. The construction of chiral tetrasubstituted carbon centers bearing trifluoromethylated N,S-ketals has been achieved in high yields (up to 96% yield) with excellent enantioselectivities (up to 99% ee).
中文翻译:

硫醇对三氟甲基酮亚胺的对映选择性加成:N,S-缩酮的合成。
通过使用衍生自奎宁的双功能方甲酰胺催化剂,建立了硫醇对无环三氟甲基酮亚胺的高效对映选择性加成反应,反应在 5 至 10 分钟内完成。已经以高产率(高达 96% 的产率)和优异的对映选择性(高达 99% ee)实现了手性四取代碳中心的构建,这些碳中心带有三氟甲基化的N,S-缩酮。
更新日期:2020-09-30
中文翻译:

硫醇对三氟甲基酮亚胺的对映选择性加成:N,S-缩酮的合成。
通过使用衍生自奎宁的双功能方甲酰胺催化剂,建立了硫醇对无环三氟甲基酮亚胺的高效对映选择性加成反应,反应在 5 至 10 分钟内完成。已经以高产率(高达 96% 的产率)和优异的对映选择性(高达 99% ee)实现了手性四取代碳中心的构建,这些碳中心带有三氟甲基化的N,S-缩酮。









































 京公网安备 11010802027423号
京公网安备 11010802027423号