Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
The reduction performance of double bonds regulated by the competition of push–pull electron groups to realize the colorimetric and fluorescence recognition of hypochlorous acid
Analyst ( IF 3.6 ) Pub Date : 2020-08-27 , DOI: 10.1039/d0an01551b Yan Shi 1, 2, 3, 4, 5 , Fangjun Huo 3, 4, 5, 6 , Yongbin Zhang 3, 4, 5, 6 , Caixia Yin 1, 2, 3, 4, 5
Analyst ( IF 3.6 ) Pub Date : 2020-08-27 , DOI: 10.1039/d0an01551b Yan Shi 1, 2, 3, 4, 5 , Fangjun Huo 3, 4, 5, 6 , Yongbin Zhang 3, 4, 5, 6 , Caixia Yin 1, 2, 3, 4, 5
Affiliation
|
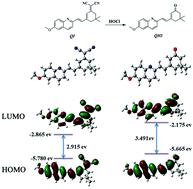
Based on its reducibility, the double bond can act as a reaction site for hypochlorous acid (HOCl), which had been demonstrated by a great deal of work. Nevertheless, the reactivity is influenced by the adjacent chemical environment. Therefore, in this work, we constructed a probe (QI) by methoxy-substituted quinoline conjugating dicyanoisoflurone, in which dicyano and pyridine N act as electron-withdrawing groups and the methoxy acts as an electron-donating group, to regulate their adjacent C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C reactivity. The “push–pull” electron effect between the methoxy group and the pyridine N led to the C
C reactivity. The “push–pull” electron effect between the methoxy group and the pyridine N led to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond being passivated. On the other hand, another C
C bond being passivated. On the other hand, another C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond was activated by the strong electron-pulling effect of the dicyano group. Thus, the previously weak intramolecular charge transfer became stronger after the dicyano adjacent to the C
C bond was activated by the strong electron-pulling effect of the dicyano group. Thus, the previously weak intramolecular charge transfer became stronger after the dicyano adjacent to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C was oxidized by HOCl, and showed a strong emission shifted from 570 to 520 nm along with a color change. The reaction mechanism was verified by mass spectrometry, NMR and theoretical calculation, and further bioimaging demonstrated the practical application of the probe.
C was oxidized by HOCl, and showed a strong emission shifted from 570 to 520 nm along with a color change. The reaction mechanism was verified by mass spectrometry, NMR and theoretical calculation, and further bioimaging demonstrated the practical application of the probe.
中文翻译:

双键的还原性能受推挽式电子基团竞争的调节,从而实现次氯酸的比色和荧光识别
基于其可还原性,双键可以充当次氯酸(HOCl)的反应位点,这已被大量工作证明。然而,反应性受邻近的化学环境影响。因此,在这项工作中,我们构建了一个由甲氧基取代的喹啉与双氰基异氟隆共轭的探针(QI),其中双氰基和吡啶N充当吸电子基团,甲氧基充当供电子基团,以调节其相邻的C![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C。反应性。甲氧基和吡啶N之间的“推挽”电子效应导致C
C。反应性。甲氧基和吡啶N之间的“推挽”电子效应导致C ![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C键被钝化。另一方面,另一个C
C键被钝化。另一方面,另一个C![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C键被双氰基的强电子拉动作用激活。因此,先前较弱的分子内电荷转移在与CC相邻的二氰基
C键被双氰基的强电子拉动作用激活。因此,先前较弱的分子内电荷转移在与CC相邻的二氰基![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) 被HOCl氧化后变得更强,并且显示出强发射从570nm转移至520nm并伴随颜色变化。通过质谱,核磁共振和理论计算验证了反应机理,进一步的生物成像证明了该探针的实际应用。
被HOCl氧化后变得更强,并且显示出强发射从570nm转移至520nm并伴随颜色变化。通过质谱,核磁共振和理论计算验证了反应机理,进一步的生物成像证明了该探针的实际应用。
更新日期:2020-09-08
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C reactivity. The “push–pull” electron effect between the methoxy group and the pyridine N led to the C
C reactivity. The “push–pull” electron effect between the methoxy group and the pyridine N led to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond being passivated. On the other hand, another C
C bond being passivated. On the other hand, another C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond was activated by the strong electron-pulling effect of the dicyano group. Thus, the previously weak intramolecular charge transfer became stronger after the dicyano adjacent to the C
C bond was activated by the strong electron-pulling effect of the dicyano group. Thus, the previously weak intramolecular charge transfer became stronger after the dicyano adjacent to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C was oxidized by HOCl, and showed a strong emission shifted from 570 to 520 nm along with a color change. The reaction mechanism was verified by mass spectrometry, NMR and theoretical calculation, and further bioimaging demonstrated the practical application of the probe.
C was oxidized by HOCl, and showed a strong emission shifted from 570 to 520 nm along with a color change. The reaction mechanism was verified by mass spectrometry, NMR and theoretical calculation, and further bioimaging demonstrated the practical application of the probe.
中文翻译:

双键的还原性能受推挽式电子基团竞争的调节,从而实现次氯酸的比色和荧光识别
基于其可还原性,双键可以充当次氯酸(HOCl)的反应位点,这已被大量工作证明。然而,反应性受邻近的化学环境影响。因此,在这项工作中,我们构建了一个由甲氧基取代的喹啉与双氰基异氟隆共轭的探针(QI),其中双氰基和吡啶N充当吸电子基团,甲氧基充当供电子基团,以调节其相邻的C
![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C。反应性。甲氧基和吡啶N之间的“推挽”电子效应导致C
C。反应性。甲氧基和吡啶N之间的“推挽”电子效应导致C ![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C键被钝化。另一方面,另一个C
C键被钝化。另一方面,另一个C![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) C键被双氰基的强电子拉动作用激活。因此,先前较弱的分子内电荷转移在与CC相邻的二氰基
C键被双氰基的强电子拉动作用激活。因此,先前较弱的分子内电荷转移在与CC相邻的二氰基![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) 被HOCl氧化后变得更强,并且显示出强发射从570nm转移至520nm并伴随颜色变化。通过质谱,核磁共振和理论计算验证了反应机理,进一步的生物成像证明了该探针的实际应用。
被HOCl氧化后变得更强,并且显示出强发射从570nm转移至520nm并伴随颜色变化。通过质谱,核磁共振和理论计算验证了反应机理,进一步的生物成像证明了该探针的实际应用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号