当前位置:
X-MOL 学术
›
ACS Med. Chem. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Facile Synthesis of Aminomethyl Phosphinate Esters as Serine Protease Inhibitors with Primed Site Interaction.
ACS Medicinal Chemistry Letters ( IF 3.5 ) Pub Date : 2020-08-10 , DOI: 10.1021/acsmedchemlett.0c00284 Jan Pascal Kahler 1 , Stijn Lenders 1 , Merel A T van de Plassche 1 , Steven H L Verhelst 1, 2
ACS Medicinal Chemistry Letters ( IF 3.5 ) Pub Date : 2020-08-10 , DOI: 10.1021/acsmedchemlett.0c00284 Jan Pascal Kahler 1 , Stijn Lenders 1 , Merel A T van de Plassche 1 , Steven H L Verhelst 1, 2
Affiliation
|
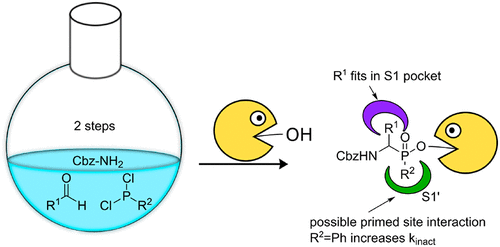
Serine proteases comprise about one-third of all proteases, and defective regulation of serine proteases is involved in numerous diseases. Therefore, serine protease inhibitors are promising drug candidates. Aminomethyl diphenyl phosphonates have been regularly used as scaffolds for covalent serine protease inhibition and the design of activity-based probes. However, they cannot make use of a protease’s primed site. Therefore, we developed a facile two-step synthesis toward a set of phenyl phosphinates, which is a related scaffold but can interact with the primed site. We tested their inhibitory activity on five different serine proteases and found that a phenyl group directly attached to the phosphorus atom leads to superior activity compared with phosphonates.
中文翻译:

作为丝氨酸蛋白酶抑制剂的氨基甲基次膦酸酯的简易合成与引发位点相互作用。
丝氨酸蛋白酶约占所有蛋白酶的三分之一,并且丝氨酸蛋白酶的缺陷调节涉及许多疾病。因此,丝氨酸蛋白酶抑制剂是有前景的候选药物。氨基甲基二苯基膦酸盐经常被用作共价丝氨酸蛋白酶抑制和基于活性的探针设计的支架。然而,它们不能利用蛋白酶的引发位点。因此,我们针对一组苯基次膦酸盐开发了一种简便的两步合成法,这是一种相关的支架,但可以与引发位点相互作用。我们测试了它们对五种不同丝氨酸蛋白酶的抑制活性,发现与膦酸盐相比,直接连接到磷原子上的苯基具有更高的活性。
更新日期:2020-09-10
中文翻译:

作为丝氨酸蛋白酶抑制剂的氨基甲基次膦酸酯的简易合成与引发位点相互作用。
丝氨酸蛋白酶约占所有蛋白酶的三分之一,并且丝氨酸蛋白酶的缺陷调节涉及许多疾病。因此,丝氨酸蛋白酶抑制剂是有前景的候选药物。氨基甲基二苯基膦酸盐经常被用作共价丝氨酸蛋白酶抑制和基于活性的探针设计的支架。然而,它们不能利用蛋白酶的引发位点。因此,我们针对一组苯基次膦酸盐开发了一种简便的两步合成法,这是一种相关的支架,但可以与引发位点相互作用。我们测试了它们对五种不同丝氨酸蛋白酶的抑制活性,发现与膦酸盐相比,直接连接到磷原子上的苯基具有更高的活性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号