Catalysis Today ( IF 5.2 ) Pub Date : 2020-08-02 , DOI: 10.1016/j.cattod.2020.07.049 Yang Yu , Zhoufeng Bian , Zhigang Wang , Juan Wang , Wenyi Tan , Qin Zhong , Sibudjing Kawi
|
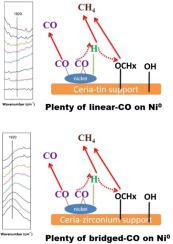
The effect of different cations (M=Zr, Sn or Ti) doping on ceria supported nickel (Ni-Ce0.8M0.2O2) catalysts and their performance in the CO2 methanation reaction has been investigated. Among the prepared catalysts, the Ni-CeO2 (CN) catalyst exhibited the best catalytic performance with a CO2 conversion of 62.9% at 400 °C. The selectivity on Ni-Ce0.8Zr0.2O2 (CZN) remained above 98.4% from 200 to 400 °C. As confirmed by in-situ DRIFTS, the dissociative adsorption of H2 mainly proceeds on Ni, by-product CO is mainly assigned to the releasing of weak-binding linearly adsorbed CO on nickel, which is from the hydrogenation of formates with adsorbed H. The CO-suppressing effect is attributed to the strong adsorption of carbon monoxide on nickel via the forming of bridging carbonyls. It was found that CO2 is adsorbed on the catalysts to form carbonate, bicarbonate and formate species. The formate species could be further hydrogenated by dissociated H and then finally release methane.
中文翻译:

Ni-Ce 0.8 M 0.2 O 2(M = Zr,Sn或Ti)催化剂上的CO 2甲烷化:通过在镍上形成桥连羰基来抑制CO
研究了不同阳离子(M = Zr,Sn或Ti)掺杂对二氧化铈负载的镍(Ni-Ce 0.8 M 0.2 O 2)催化剂的影响及其在CO 2甲烷化反应中的性能。在制备的催化剂中,Ni-CeO 2(CN)催化剂表现出最佳的催化性能,在400°C下的CO 2转化率为62.9%。从200到400°C ,Ni-Ce 0.8 Zr 0.2 O 2(CZN)的选择性保持在98.4%以上。如原位DRIFTS所证实的,H 2的解离吸附副产物CO主要是由镍吸附形成的甲酸酯加氢而产生的。CO的抑制作用归因于一氧化碳的强吸附通过桥连羰基的形成在镍上 发现CO 2吸附在催化剂上形成碳酸盐,碳酸氢盐和甲酸盐种类。甲酸盐可以通过离解的H进一步氢化,然后最终释放出甲烷。











































 京公网安备 11010802027423号
京公网安备 11010802027423号