Acta Pharmaceutica Sinica B ( IF 14.7 ) Pub Date : 2020-07-30 , DOI: 10.1016/j.apsb.2020.07.015 Qiaozhen Wang 1 , Xiaolong Tang 2 , Ping Dai 1 , Chuanxi Wang 1 , Weiyang Zhang 2 , Guodong Chen 1 , Kui Hong 3 , Dan Hu 1 , Hao Gao 1 , Xinsheng Yao 1
|
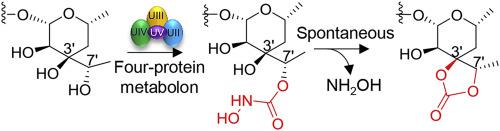
Organic carbonates (OCs) are a class of compounds featured by a carbonyl flanked by two alkoxy/aryloxy groups. They exist in either linear or cyclic forms, of which the majority encountered in nature adopt a pentacyclic structure. However, the enzymatic basis for pentacyclic carbonate ring formation remains elusive. Here, we reported that a four-protein metabolon (AlmUII–UV) assembled by a small peptide protein (AlmUV) appends a reactive N-hydroxylcarbamoyl moiety to the decarboxylated aldgamycins followed by a non-enzymatic condensation to give the pentacyclic carbonate ring. Our results have documented an unprecedent mechanism for carbonate formation.
中文翻译:

由小肽蛋白组装的四蛋白代谢子产生阿尔加霉素的五环碳酸酯环
有机碳酸酯 (OC) 是一类以羰基为特征的化合物,其两侧为两个烷氧基/芳氧基。它们以线性或环状形式存在,其中自然界中遇到的大多数采用五环结构。然而,五环碳酸酯环形成的酶学基础仍然难以捉摸。在这里,我们报道了由小肽蛋白 (AlmUV) 组装的四蛋白代谢子 (AlmUII-UV) 将反应性N-羟基氨基甲酰基部分附加到脱羧藻霉素上,然后进行非酶缩合得到五环碳酸酯环。我们的结果记录了碳酸盐形成的前所未有的机制。










































 京公网安备 11010802027423号
京公网安备 11010802027423号