当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Cobalt-Catalyzed Selective Unsymmetrical Dioxidation of gem-Difluoroalkenes.
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-07-22 , DOI: 10.1021/acs.joc.0c00415 Douglas L Orsi 1 , Justin T Douglas 2 , Jacob P Sorrentino 3 , Ryan A Altman 4, 5
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-07-22 , DOI: 10.1021/acs.joc.0c00415 Douglas L Orsi 1 , Justin T Douglas 2 , Jacob P Sorrentino 3 , Ryan A Altman 4, 5
Affiliation
|
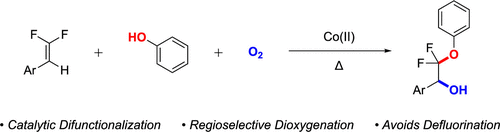
gem-Difluoroalkenes represent valuable synthetic handles for organofluorine chemistry; however, most reactions of this substructure proceed through reactive intermediates prone to eliminate a fluorine atom and generate monofluorinated products. Taking advantage of the distinct reactivity of gem-difluoroalkenes, we present a cobalt-catalyzed regioselective unsymmetrical dioxygenation of gem-difluoroalkenes using phenols and molecular oxygen, which retains both fluorine atoms and provides β-phenoxy-β,β-difluorobenzyl alcohols. Mechanistic studies suggest that the reaction operates through a radical chain process initiated by Co(II)/O2/phenol and quenched by the Co-based catalyst. This mechanism enables the retention of both fluorine atoms, which contrasts most transition-metal-catalyzed reactions of gem-difluoroalkenes that typically involve defluorination.
中文翻译:

钴催化的 gem-二氟烯烃的选择性不对称二氧化。
gem -二氟烯烃代表了有机氟化学的宝贵合成手柄;然而,该子结构的大多数反应通过易于消除氟原子并生成单氟化产物的反应中间体进行。取的不同反应性的优点宝石-difluoroalkenes中,我们提出的钴催化的区域选择性不对称双加氧宝石-difluoroalkenes使用酚和分子氧,它保留两个氟原子,并提供β苯氧基- β,β-二氟苄基醇。机理研究表明,该反应通过由 Co(II)/O 2引发的自由基链过程进行/苯酚并被钴基催化剂淬灭。这种机制能够保留两个氟原子,这与通常涉及脱氟的大多数过渡金属催化的鎓二氟烯烃反应形成对比。
更新日期:2020-08-21
中文翻译:

钴催化的 gem-二氟烯烃的选择性不对称二氧化。
gem -二氟烯烃代表了有机氟化学的宝贵合成手柄;然而,该子结构的大多数反应通过易于消除氟原子并生成单氟化产物的反应中间体进行。取的不同反应性的优点宝石-difluoroalkenes中,我们提出的钴催化的区域选择性不对称双加氧宝石-difluoroalkenes使用酚和分子氧,它保留两个氟原子,并提供β苯氧基- β,β-二氟苄基醇。机理研究表明,该反应通过由 Co(II)/O 2引发的自由基链过程进行/苯酚并被钴基催化剂淬灭。这种机制能够保留两个氟原子,这与通常涉及脱氟的大多数过渡金属催化的鎓二氟烯烃反应形成对比。









































 京公网安备 11010802027423号
京公网安备 11010802027423号