European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2020-07-07 , DOI: 10.1016/j.ejmech.2020.112554 Nerea Escala , Elora Valderas-García , María Álvarez Bardón , Verónica Castilla Gómez de Agüero , Ricardo Escarcena , José Luis López-Pérez , Francisco A. Rojo-Vázquez , Arturo San Feliciano , Rafael Balaña-Fouce , María Martínez-Valladares , Esther del Olmo
|
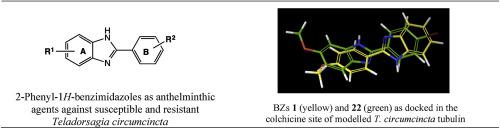
Gastrointestinal nematode infections are the main diseases in herds of small ruminants. Resistance to the main established drugs has become a worldwide problem. The purpose of this study is to obtain and evaluate the in vitro ovicidal and larvicidal activity of some 2-phenylbenzimidazole derivatives on susceptible and resistant strains of Teladorsagia circumcincta. Compounds were prepared by known procedures from substituted o-phenylenediamines and arylaldehydes or intermediate sodium 1-hydroxyphenylmethanesulfonate derivatives. Egg Hatch Test (EHT), Larval Mortality Test (LMT) and Larval Migration Inhibition Test (LMIT) were used in the initial screening of compounds at 50 μM concentration, and EC50 values were determined for the most potent compounds. Cytotoxicity evaluation of compounds was conducted on human Caco-2 and HepG2 cell lines to calculate their Selectivity Indexes (SI). At 50 μM concentration, nine out of twenty-four compounds displayed more than 98% ovicidal activity on a susceptible strain, and four of them showed more than 86% on one resistant strain. The most potent ovicidal benzimidazole (BZ) 3 showed EC50 = 6.30 μM, for the susceptible strain, while BZ 2 showed the lowest EC50 value of 14.5 μM for the resistant strain. Docking studies of most potent compounds in a modelled Teladorsagia tubulin indicated an inverted orientation for BZ 1 in the colchicine binding site, probably due to its fair interaction with glutamic acid at codon 198, which could justify its inactivity against the resistant strain of T. circumcincta.
中文翻译:

某些2-苯基-1 H-苯并咪唑衍生物作为驱虫药抗线虫Teladorsagia circumcincta的合成,生物评价和对接研究
胃肠道线虫感染是小反刍动物群中的主要疾病。对主要已确立的药物的耐药性已成为世界性问题。本研究的目的是获得和评价某些2-苯基苯并咪唑衍生物对环柏变易感菌株的体外杀卵和杀幼虫活性。通过已知方法由取代的邻苯二胺和芳醛或中间体1-羟基苯基甲磺酸钠衍生物制备化合物。卵孵化试验(EHT),幼虫死亡率试验(LMT)和幼虫迁移抑制试验(LMIT)用于浓度为50μM和EC 50的化合物的初始筛选确定最有效化合物的值。在人Caco-2和HepG2细胞系上进行了化合物的细胞毒性评估,以计算其选择性指数(SI)。在50μM浓度下,二十四个化合物中的九个对易感菌株显示出超过98%的杀卵活性,其中四个对一种抗性菌株显示出超过86%的杀卵活性。 对于易感菌株,最有效的杀卵剂苯并咪唑(BZ)3的EC 50 = 6.30μM,而对于抗性菌株,BZ 2的EC 50值最低,为14.5μM。在模拟的Teladorsagia微管蛋白中对大多数有效化合物的对接研究表明BZ 1的方向相反在秋水仙碱的结合位点,可能是由于它与第198位密码子的谷氨酸有公平的相互作用,这可能证明它对环柏的抗药性没有活性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号