Water Research ( IF 12.8 ) Pub Date : 2020-07-02 , DOI: 10.1016/j.watres.2020.116116 Zhi Liu , Bin Xu , Yi-Li Lin , Tian-Yang Zhang , Tao Ye , Chen-Yan Hu , Yong-Shan Lu , Tong-Cheng Cao , Yu-Lin Tang , Nai-Yun Gao
|
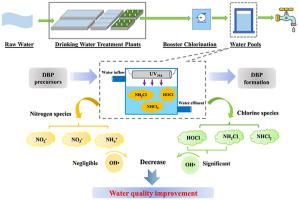
The conversion mechanisms of chlorine species (including free chlorine, monochloramine (NH2Cl), dichloramine, and total chlorine), nitrogen species (including ammonium (NH4+), nitrate (NO3−), and nitrite (NO2−)) as well as the formation of disinfection by-products (DBPs) in a UV-activated mixed chlorine/chloramines system in water were investigated in this work. The consumption rates of free chlorine and NH2Cl were significantly promoted in a HOCl/NH2Cl coexisting system, especially in the presence of UV irradiation. Moreover, the transformation forms of nitrogen in both ultrapure and HA-containing waters were considerably affected by UV irradiation and the mass ratio of free chlorine to NH2Cl. NO3− and NO2− can be easily produced under UV irradiation, and the removal efficiency of total nitrogen with UV was obvious higher than that without UV when the initial ratio of HOCl/NH2Cl was less than 1. The roles of different radicals in the degradation of free chlorine, NH2Cl and NH4+ were also considered in such a UV-activated mixed chlorine/chloramines system. The results indicated that OH• was important to the consumption of free chlorine and NH2Cl, and showed negligible influence on the consumption of NH4+. Besides, the changes of DOC and UV254 in HA-containing water in UV-activated mixed chlorine/chloramines system indicated that the removal efficiency of DOC (24%) was much lower than that of UV254 (94%). The formation of DBPs in a mixed chlorine/chloramines system was also evaluated. The yields of DBPs decreased significantly as the mass ratio of HOCl/NH2Cl varied from 1 : 0 to 0 : 1. Moreover, compared to the conditions without UV irradiation, higher DBPs yields and DBP-associated calculated toxicity were observed during the UV-activated mixed chlorine/chloramine process.
中文翻译:

在紫外线活化的氯/氯胺混合系统中氯/氮转化和消毒副产物生成的机理研究。
氯物质(包括游离氯,氯胺(NH的转换机构2 Cl)的,二氯胺,和总氯),氮物种(包括铵(NH 4 +),硝酸(NO 3 - ),和亚硝酸根(NO 2 - )在这项工作中,还研究了紫外线活化的水中氯/氯胺混合系统中消毒副产物(DBP)的形成。在HOCl / NH 2中,游离氯和NH 2 Cl的消耗速率显着提高Cl共存体系,特别是在紫外线照射下。此外,紫外线辐射和游离氯与NH 2 Cl的质量比极大地影响了超纯水和含HA的水中氮的转化形式。NO 3 -和NO 2 -可以很容易地在紫外光照射下产生的,并用UV总氮的去除率比明显高于无UV当次氯酸/ NH的初始比例2氯是小于1的不同的作用自由基降解游离氯,NH 2 Cl和NH 4 +在这种紫外线活化的氯/氯胺混合体系中也考虑了这一点。结果表明,OH·对游离氯和NH 2 Cl的消耗很重要,对NH 4 +的消耗影响可忽略不计。此外,紫外线活化氯/氯胺混合体系中含HA的水中DOC和UV 254的变化表明,DOC的去除效率(24%)远低于UV 254(94%)。还评估了氯/氯胺混合体系中DBP的形成。随着HOCl / NH 2的质量比,DBPs的产率显着下降。Cl在1∶0至0∶1之间变化。此外,与未进行UV照射的条件相比,在UV活化的氯/氯胺混合过程中观察到更高的DBP产率和与DBP相关的计算毒性。



























 京公网安备 11010802027423号
京公网安备 11010802027423号