当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Enantioselective Synthesis of Functionalized Diazaspirocycles from 4‐Benzylideneisoxazol‐5(4H)‐one Derivatives and Isocyanoacetate Esters
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-06-29 , DOI: 10.1002/adsc.202000611 Pablo Martínez‐Pardo 1 , Adrián Laviós 1 , Amparo Sanz‐Marco 1 , Carlos Vila 1 , José R. Pedro 1 , Gonzalo Blay 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-06-29 , DOI: 10.1002/adsc.202000611 Pablo Martínez‐Pardo 1 , Adrián Laviós 1 , Amparo Sanz‐Marco 1 , Carlos Vila 1 , José R. Pedro 1 , Gonzalo Blay 1
Affiliation
|
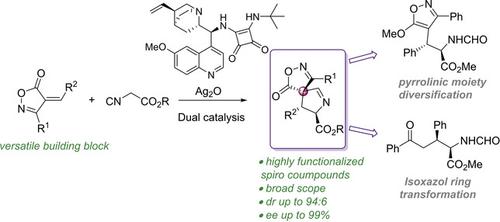
Enantioenriched spirocyclic compounds bearing three contiguous stereocenters and high functionalization were obtained through a formal [3+2] cycloaddition reaction catalyzed by a cooperative system. The spiro compounds were synthesized from 4‐arylideneisoxazol‐5‐ones and isocyanoacetate esters using a bifunctional squaramide/Brønsted base organocatalyst derived from a Cinchona alkaloid and silver oxide as Lewis acid. This method afforded two out of the four possible diastereomers with good yields and high enantiomeric excess for both diastereomers.
中文翻译:

从4-苄叉基异恶唑-5(4H)-one衍生物和异氰酸酯基酯的对映选择性合成功能性二氮杂螺环
通过协同系统催化的正式的[3 + 2]环加成反应,获得了具有三个连续立体中心和高官能度的对映体富集的螺环化合物。使用由金鸡纳生物碱和氧化银(路易斯酸)衍生的双官能方胺/布朗斯台德碱有机催化剂,由4-芳叉基异恶唑5-1和异氰基乙酸酯合成螺化合物。该方法提供了四种可能的非对映异构体中的两种,具有良好的收率和两种非对映异构体的高对映体过量。
更新日期:2020-09-05
中文翻译:

从4-苄叉基异恶唑-5(4H)-one衍生物和异氰酸酯基酯的对映选择性合成功能性二氮杂螺环
通过协同系统催化的正式的[3 + 2]环加成反应,获得了具有三个连续立体中心和高官能度的对映体富集的螺环化合物。使用由金鸡纳生物碱和氧化银(路易斯酸)衍生的双官能方胺/布朗斯台德碱有机催化剂,由4-芳叉基异恶唑5-1和异氰基乙酸酯合成螺化合物。该方法提供了四种可能的非对映异构体中的两种,具有良好的收率和两种非对映异构体的高对映体过量。











































 京公网安备 11010802027423号
京公网安备 11010802027423号