European Journal of Medicinal Chemistry ( IF 6.7 ) Pub Date : 2020-06-27 , DOI: 10.1016/j.ejmech.2020.112513 Aya A M Mohammed 1 , Ghadeer A R Y Suaifan 1 , Mayadah B Shehadeh 1 , Patrick N Okechukwu 2
|
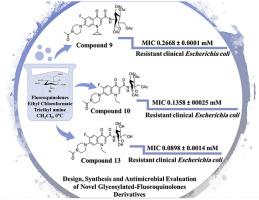
Herein we report the design, synthesis and biological evaluation of structurally modified ciprofloxacin, norfloxacin and moxifloxacin standard drugs, featuring amide functional groups at C-3 of the fluoroquinolone scaffold. In vitro antimicrobial testing against various Gram-positive bacteria, Gram-negative bacteria and fungi revealed potential antibacterial and antifungal activity. Hybrid compounds 9 (MIC 0.2668 ± 0.0001 mM), 10 (MIC 0.1358 ± 00025 mM) and 13 (MIC 0.0898 ± 0.0014 mM) had potential antimicrobial activity against a fluoroquinolone-resistant Escherichia coli clinical isolate, compared to ciprofloxacin (MIC 0.5098 ± 0.0024 mM) and norfloxacin (MIC 0.2937 ± 0.0021 mM) standard drugs. Interestingly, compound 10 also exerted potential antifungal activity against Candida albicans (MIC 0.0056 ± 0.0014 mM) and Penicillium chrysogenum (MIC 0.0453 ± 0.0156 mM). Novel derivatives and standard fluoroquinolone drugs exhibited near-identical cytotoxicity levels against L6 muscle cell-line, when measured using the MTT assay.
中文翻译:

新型糖基化氟喹诺酮衍生物的设计,合成和抗菌评估。
本文中,我们报告了结构修饰的环丙沙星,诺氟沙星和莫西沙星标准药物的设计,合成和生物学评估,这些药物在氟喹诺酮骨架的C-3处具有酰胺官能团。对各种革兰氏阳性细菌,革兰氏阴性细菌和真菌的体外抗菌测试显示出潜在的抗菌和抗真菌活性。杂合化合物9(MIC 0.2668±0.0001 mM),10(MIC 0.1358±00025 mM)和13(MIC 0.0898±0.0014 mM)对耐氟喹诺酮类大肠杆菌具有潜在的抗菌活性与环丙沙星(MIC 0.5098±0.0024 mM)和诺氟沙星(MIC 0.2937±0.0021 mM)标准药物相比。有趣的是,化合物10还对白色念珠菌(MIC 0.0056±0.0014 mM)和产黄青霉(MIC 0.0453±0.0156 mM)发挥潜在的抗真菌活性。当使用MTT测定法测量时,新型衍生物和标准的氟喹诺酮类药物对L6肌肉细胞系表现出几乎相同的细胞毒性水平。



























 京公网安备 11010802027423号
京公网安备 11010802027423号