Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Impact of Molecular Shape on Supramolecular Copolymer Synthesis in Seeded Living Polymerization of Perylene Bisimides
CCS Chemistry ( IF 9.4 ) Pub Date : 2019-12-09 , DOI: 10.31635/ccschem.019.20190061 Wolfgang Wagner 1, 2 , Marius Wehner 1, 2 , Vladimir Stepanenko 2 , Frank Würthner 1, 2
CCS Chemistry ( IF 9.4 ) Pub Date : 2019-12-09 , DOI: 10.31635/ccschem.019.20190061 Wolfgang Wagner 1, 2 , Marius Wehner 1, 2 , Vladimir Stepanenko 2 , Frank Würthner 1, 2
Affiliation
|
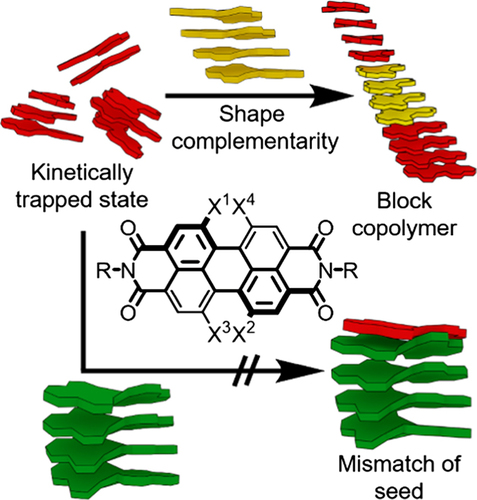
Supramolecular polymerization properties have been
studied for a series of perylene bisimide (PBI) dyes
containing identical hydrogen-bonding amide groups
in imide positions but variable number or size of
alkoxy substituents in bay-positions. Temperatureand time-dependent UV/vis and atomic force microscopy (AFM) studies revealed that the number and
steric demand of the alkoxy substituents in baypositions has a strong impact on thermodynamically,
and more importantly, on kinetically controlled supramolecular polymerization of these PBIs in solvent
mixtures of methylcyclohexane and toluene. Our studies revealed that in the case of core tri- and tetramethoxy-substituted PBIs, the kinetically controlled
aggregation pathway was suppressed completely,
whereas monomethoxy and 1,7-dialkoxy PBI derivatives form kinetically trapped species. Besides, the
monomethoxy-substituted PBI could be used successfully in self-seeded chain-growth polymerization
of monomeric building blocks under precisely kinetically controlled conditions to obtain supramolecular
polymers with controlled length and low polydispersity. Further, two-component seed-induced living
copolymerization of the present PBI series revealed
that the formation of supramolecular block copolymers is only feasible for appropriate combinations
of PBIs and that the seeding efficiency is strongly
dependent on the shape complementarity of the individual components applied. Thus, seeded supramolecular polymerization and copolymerization of PBIs
could be modulated by subtle changes of the molecular shape through rational design of monomers.
中文翻译:

sim双酰亚胺种子活性聚合中分子形状对超分子共聚物合成的影响
已经研究了一系列在酰亚胺位置包含相同的氢键合酰胺基团但在海湾位置具有可变数目或大小的烷氧基取代基的per双酰亚胺(PBI)染料。温度和时间相关的UV / vis和原子力显微镜(AFM)研究表明,烷氧基取代基的数目和空间需求对热力学,尤其是对动力学的控制有很大影响。这些PBI在甲基环己烷和甲苯的溶剂混合物中的分子聚合。我们的研究表明,在核心三和四甲氧基取代的PBI的情况下,动力学控制的聚集途径被完全抑制,而单甲氧基和1,7-二烷氧基PBI衍生物形成了动力学捕获的物种。除了,单甲氧基取代的PBI可以成功地在精确的运动控制条件下用于单体结构单元的自种链增长聚合中,从而获得具有可控制的长度和低多分散性的超分子聚合物。此外,本PBI系列的两组分种子诱导的活性共聚表明,超分子嵌段共聚体的形成仅适用于PBI的适当组合,且播种效率强烈取决于个体的形状互补性应用的组件。因此,通过合理设计单体,可以通过分子形状的细微变化来调节PBI的种子超分子聚合和共聚。
更新日期:2020-06-24
中文翻译:

sim双酰亚胺种子活性聚合中分子形状对超分子共聚物合成的影响
已经研究了一系列在酰亚胺位置包含相同的氢键合酰胺基团但在海湾位置具有可变数目或大小的烷氧基取代基的per双酰亚胺(PBI)染料。温度和时间相关的UV / vis和原子力显微镜(AFM)研究表明,烷氧基取代基的数目和空间需求对热力学,尤其是对动力学的控制有很大影响。这些PBI在甲基环己烷和甲苯的溶剂混合物中的分子聚合。我们的研究表明,在核心三和四甲氧基取代的PBI的情况下,动力学控制的聚集途径被完全抑制,而单甲氧基和1,7-二烷氧基PBI衍生物形成了动力学捕获的物种。除了,单甲氧基取代的PBI可以成功地在精确的运动控制条件下用于单体结构单元的自种链增长聚合中,从而获得具有可控制的长度和低多分散性的超分子聚合物。此外,本PBI系列的两组分种子诱导的活性共聚表明,超分子嵌段共聚体的形成仅适用于PBI的适当组合,且播种效率强烈取决于个体的形状互补性应用的组件。因此,通过合理设计单体,可以通过分子形状的细微变化来调节PBI的种子超分子聚合和共聚。











































 京公网安备 11010802027423号
京公网安备 11010802027423号