Chem ( IF 19.1 ) Pub Date : 2020-06-23 , DOI: 10.1016/j.chempr.2020.05.017 Zijun Zhou , Yuqi Tan , Tatsuya Yamahira , Sergei Ivlev , Xiulan Xie , Radostan Riedel , Marcel Hemming , Masanari Kimura , Eric Meggers
|
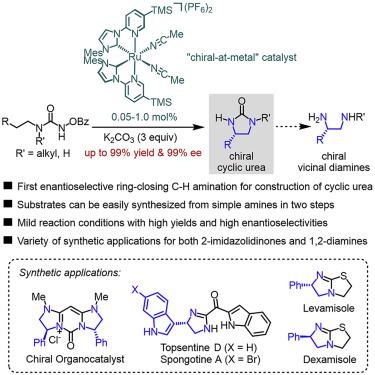
An enantioselective intramolecular C(sp3)–H amination of N-benzoyloxyurea by using a chiral-at-metal ruthenium catalyst is reported, providing chiral 2-imidazolidinones in yields of up to 99% and with up to 99% ee. Catalyst loadings down to 0.05 mol % are feasible. Control experiments support a stepwise nitrene insertion mechanism through hydrogen atom transfer of a ruthenium nitrenoid intermediate followed by a radical recombination. Chiral 2-imidazolidinones are prevalent in bioactive compounds and can be converted to chiral vicinal diamines in a single step. The synthetic value of the new method is demonstrated for the synthesis of intermediates of the drugs levamisole and dexamisole, the bisindole alkaloids topsentine D and spongotine A, and a chiral organocatalyst.
中文翻译:

尿素衍生物的对映选择性环封闭式C–H胺化
据报道,通过使用手性金属钌催化剂进行N-苯甲酰氧基脲的对映选择性分子内C(sp 3)-H胺化反应,可提供手性2-咪唑烷酮类,产率高达99%,ee高达99%。降低至0.05mol%的催化剂负载是可行的。对照实验通过钌氮烯类中间体的氢原子转移然后进行自由基重组来支持逐步的氮烯插入机理。手性2-咪唑啉酮在生物活性化合物中普遍存在,并且可以一步转化为手性邻位二胺。证明了该新方法的合成价值用于合成药物左旋咪唑和右旋咪唑,双吲哚生物碱托普汀D和海绵蛋白A的中间体,以及手性有机催化剂。









































 京公网安备 11010802027423号
京公网安备 11010802027423号