当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Selective C-H chalcogenation of thiazoles via thiazol-2-yl-phosphonium salts.
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-06-17 , DOI: 10.1039/d0ob00684j You Zi 1 , Konrad Wagner 1 , Fritz Schömberg 1 , Ivan Vilotijevic 1
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-06-17 , DOI: 10.1039/d0ob00684j You Zi 1 , Konrad Wagner 1 , Fritz Schömberg 1 , Ivan Vilotijevic 1
Affiliation
|
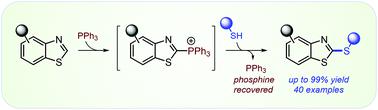
Thiazoles and benzothiazoles undergo regioselective C2–H chalcogenation via the sequence of thiazole C2-functionalization with phosphines to produce phosphonium salts which in turn react with S- and Se-centered nucleophiles to give products of C2–H chalcogenation and allow for recovery of the starting phosphine. The atom economical sequence proceeds under mild conditions and features broad scope for both the nucleophiles (electron-rich, electron-poor, sterically hindered thiols) and the various substituted benzothiazoles. The access to the substituted medicinally relevant C2-thio benzothiazoles also enables stereoselectivity improvements in the modified Julia olefinations.
中文翻译:

噻唑-2-基-盐对噻唑的选择性CH硫族化作用。
噻唑和苯并噻唑通过噻唑的C2-官能化与膦进行序列选择性的C2-H硫族化,生成phospho盐,然后与S和Se中心的亲核试剂反应生成C2-H的硫族化产物,并可以回收起始膦。原子经济序列在温和条件下进行,并且对亲核体(富电子,贫电子,空间受阻的硫醇)和各种取代的苯并噻唑都具有广阔的范围。获得取代的与医学相关的C 2-硫代苯并噻唑的方法也使得改进的茱莉亚烯化反应中的立体选择性得以改善。
更新日期:2020-07-15
中文翻译:

噻唑-2-基-盐对噻唑的选择性CH硫族化作用。
噻唑和苯并噻唑通过噻唑的C2-官能化与膦进行序列选择性的C2-H硫族化,生成phospho盐,然后与S和Se中心的亲核试剂反应生成C2-H的硫族化产物,并可以回收起始膦。原子经济序列在温和条件下进行,并且对亲核体(富电子,贫电子,空间受阻的硫醇)和各种取代的苯并噻唑都具有广阔的范围。获得取代的与医学相关的C 2-硫代苯并噻唑的方法也使得改进的茱莉亚烯化反应中的立体选择性得以改善。











































 京公网安备 11010802027423号
京公网安备 11010802027423号