Bioorganic & Medicinal Chemistry Letters ( IF 2.5 ) Pub Date : 2020-06-13 , DOI: 10.1016/j.bmcl.2020.127351 Yana B Platonova 1 , Alexander N Volov 2 , Larisa G Tomilova 1
|
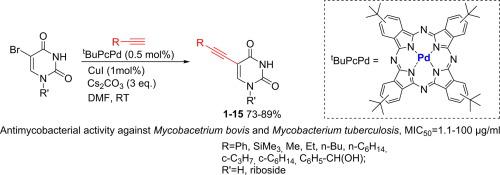
A series of new 5-alkynyl-substituted uracil and uridine derivatives were synthesised via palladium-catalysed Sonogashira cross-coupling reaction of 5-bromo-pyrimidine base with terminal acetylenes with good yields in DMF at room temperature. All obtained compounds were tested for antimycobacterial activity against Mycobacetrium bovis and Mycobacterium tuberculosis (H37Ra) at concentrations of 1–100 µg/ml using MABA test. Obtained results revealed that most of tested uracil derivatives exhibited high antimycobacterial activity (MIC50 = 1.1–19.2 µg/ml) in comparison with therapeutic agents such as rifampicin, isoniazid and d-cycloserine, excluding compounds having alkyl substituent at triple alkyne bond.
中文翻译:

5-炔基尿嘧啶衍生物的合成和抗结核活性。
在室温下,通过钯催化5-溴-嘧啶碱与末端乙炔的钯催化Sonogashira交叉偶联反应,合成了一系列新的5-炔基取代的尿嘧啶和尿苷衍生物。使用MABA试验测试了所有获得的化合物对牛分枝杆菌和结核分枝杆菌(H37Ra)的抗分枝杆菌活性(浓度为1–100 µg / ml)。所得结果表明,除 利福平,异烟肼和d-环丝氨酸等治疗剂外,大多数测试的尿嘧啶衍生物均表现出较高的抗分枝杆菌活性(MIC 50 = 1.1-19.2 µg / ml),但三炔键上具有烷基取代基的化合物除外。









































 京公网安备 11010802027423号
京公网安备 11010802027423号