当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Copper(ii)-catalyzed stereoselective 1,2-addition vs. Ferrier glycosylation of "armed" and "disarmed" glycal donors.
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-06-08 , DOI: 10.1039/d0ob01042a Manoj Kumar 1 , Thurpu Raghavender Reddy 1 , Aakanksha Gurawa 1 , Sudhir Kashyap 1
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2020-06-08 , DOI: 10.1039/d0ob01042a Manoj Kumar 1 , Thurpu Raghavender Reddy 1 , Aakanksha Gurawa 1 , Sudhir Kashyap 1
Affiliation
|
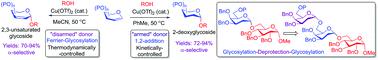
Selective activation of “armed’ and ‘'disarmed” glycal donors enabling the stereo-controlled glycosylations by employing Cu(II)-catalyst as the promoter has been realized. The distinctive stereochemical outcome in the process is mainly influenced by the presence of diverse protecting groups on the donor and the solvent system employed. The protocol is compatible with a variety of aglycones including carbohydrates, amino acids, and natural products to access deoxy-glycosides and glycoconjugates with high α-anomeric selectivity. Notably, the synthetic practicality of the method is amply verified for the stereoselective assembling of trisaccharides comprising 2-deoxy components. Mechanistic studies involving deuterated experiments validate the syn-diastereoselective 1,2-addition of acceptors on the double bond of armed donors.
中文翻译:

铜(ii)催化的立体式1,2加成与“武装”和“解除武装”的糖供体的Ferrier糖基化作用。
通过使用Cu(II)-催化剂作为启动子,实现了“武装”和“解除武装”的糖基供体的选择性激活,从而实现了立体控制的糖基化。在该方法中独特的立体化学结果主要受供体和所用溶剂体系上存在各种保护基的影响。该方案可与多种糖苷配基(包括碳水化合物,氨基酸和天然产物)兼容,以高α-异头异构体选择性获得脱氧糖苷和糖缀合物。值得注意的是,该方法的合成实用性已被充分验证用于包含2-脱氧成分的三糖的立体选择性组装。涉及氘代实验的机理研究验证了合成-在武装供体的双键上进行非对映体选择性的1,2-加成受体。
更新日期:2020-07-01
中文翻译:

铜(ii)催化的立体式1,2加成与“武装”和“解除武装”的糖供体的Ferrier糖基化作用。
通过使用Cu(II)-催化剂作为启动子,实现了“武装”和“解除武装”的糖基供体的选择性激活,从而实现了立体控制的糖基化。在该方法中独特的立体化学结果主要受供体和所用溶剂体系上存在各种保护基的影响。该方案可与多种糖苷配基(包括碳水化合物,氨基酸和天然产物)兼容,以高α-异头异构体选择性获得脱氧糖苷和糖缀合物。值得注意的是,该方法的合成实用性已被充分验证用于包含2-脱氧成分的三糖的立体选择性组装。涉及氘代实验的机理研究验证了合成-在武装供体的双键上进行非对映体选择性的1,2-加成受体。











































 京公网安备 11010802027423号
京公网安备 11010802027423号