Journal of Organometallic Chemistry ( IF 2.1 ) Pub Date : 2020-06-06 , DOI: 10.1016/j.jorganchem.2020.121356 Elena A. Saratovskikh , Vyacheslav M. Martynenko , Boris L. Psikha , Natalia A. Sanina
|
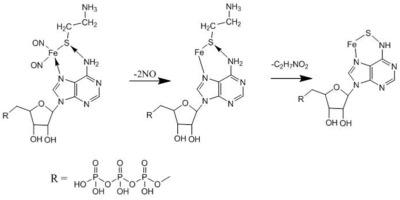
The reaction of the tetranitrosyl iron complex (nitric oxide donor) bearing the cysteamine ligand (CAC) with adenosine triphosphoric acid (ATP) was studied. The product of the reaction of CAC with ATP was isolated in the solid state and characterized by UV and IR spectroscopy. The interaction of CAC with the adenine moiety of the ATP molecule was shown using spectrofluorimetry, and the stability constant (complexation constant) of the reaction product was determined: K = (2.1 ± 0.2)∙104 mol−1 L. Ions with the mass numbers 569 and 491 m/z corresponding to [Fe–S–P3N5C10H6O12]+ and [Fe–S–P2N5C10H7O9]+ were identified in the mass spectrum of the product.
中文翻译:

腺苷三磷酸与四亚硝基铁络合物[Fe 2(S(CH 2)2 NH 3)2(NO)4 ] SO 4 ·2.5H 2 O的反应
研究了带有半胱胺配体(CAC)的四亚硝基铁络合物(一氧化氮供体)与三磷酸腺苷(ATP)的反应。CAC与ATP的反应产物以固态分离,并通过UV和IR光谱表征。使用分光荧光法显示了CAC与ATP分子的腺嘌呤部分的相互作用,并确定了反应产物的稳定性常数(络合常数):K =(2.1±0.2)∙10 4 mol -1L 。质量数569和491 m / z对应于[Fe–S–P 3 N 5 C 10 H 6 O 12 ] +和[Fe–S–P 2在产物的质谱图中鉴定出N 5 C 10 H 7 O 9 ] +。











































 京公网安备 11010802027423号
京公网安备 11010802027423号