当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Preparation of Oxindoles via Visible‐Light‐Induced Amination/Cyclization of Arylacrylamides with Alkyl Amines
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-06-02 , DOI: 10.1002/adsc.202000609 Yu‐Zhao Wang 1 , Wu‐Jie Lin 1 , Jian‐Yu Zou 1 , Wei Yu 1 , Xue‐Yuan Liu 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-06-02 , DOI: 10.1002/adsc.202000609 Yu‐Zhao Wang 1 , Wu‐Jie Lin 1 , Jian‐Yu Zou 1 , Wei Yu 1 , Xue‐Yuan Liu 1
Affiliation
|
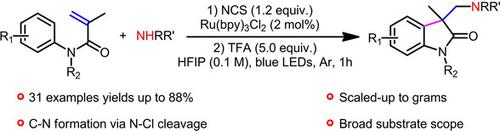
The cascade amination/cyclization of N‐arylacrylamides with alkyl amines under visible‐light photoredox catalysis is realized via intermediacy of aminium radicals. The aminium radicals are generated by a two‐step sequence which involves N‐chlorination of alkyl amines and subsequent reductive N−Cl cleavage. This method provides a convenient access to aminated oxindoles.
中文翻译:

通过可见光诱导的芳基丙烯酰胺与烷基胺的胺化/环化反应制备羟吲哚
N-芳基丙烯酰胺与烷基胺在可见光光氧化还原催化下的级联胺化/环化是通过铝自由基实现的。铝自由基是由两步序列生成的,该过程涉及烷基胺的N-氯化和随后的还原性N-Cl裂解。这种方法可以方便地获得胺化的吲哚。
更新日期:2020-08-04
中文翻译:

通过可见光诱导的芳基丙烯酰胺与烷基胺的胺化/环化反应制备羟吲哚
N-芳基丙烯酰胺与烷基胺在可见光光氧化还原催化下的级联胺化/环化是通过铝自由基实现的。铝自由基是由两步序列生成的,该过程涉及烷基胺的N-氯化和随后的还原性N-Cl裂解。这种方法可以方便地获得胺化的吲哚。











































 京公网安备 11010802027423号
京公网安备 11010802027423号