Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-05-30 , DOI: 10.1016/j.bmc.2020.115565 James M Wood 1 , Nishikant S Satam 2 , Renata G Almeida 3 , Vinicius S Cristani 4 , Dênis P de Lima 4 , Luiza Dantas-Pereira 5 , Kelly Salomão 5 , Rubem F S Menna-Barreto 5 , Irishi N N Namboothiri 2 , John F Bower 6 , Eufrânio N da Silva Júnior 3
|
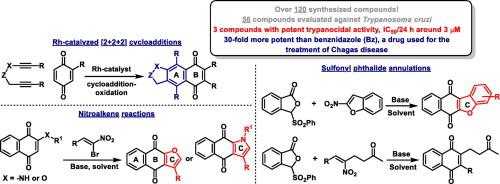
Rhodium-catalyzed [2 + 2 + 2] cycloadditions, sulfonyl phthalide annulations and nitroalkene reactions have been employed for the synthesis of 56 quinone-based compounds. These were evaluated against Trypanosoma cruzi, the parasite that causes Chagas disease. The reactions described here are part of a program that aims to utilize modern, versatile and efficient synthetic methods for the one or two step preparation of trypanocidal compounds. We have identified 9 compounds with potent activity against the parasite; 3 of these were 30-fold more potent than benznidazole (Bz), a drug used for the treatment of Chagas disease. This article provides a comprehensive outline of reactions involving over 120 compounds aimed at the discovery of new quinone-based frameworks with activity against T. cruzi.
中文翻译:

高效锥虫杀伤剂的策略:Rh催化的[2 + 2 + 2]环加成反应,磺酰邻苯二甲酸酯环化和硝基烯烃反应在取代醌的合成中的应用及其对克鲁斯锥虫的评价。
铑催化的[2 + 2 + 2]环加成反应,磺酰苯酞环化反应和硝基烯烃反应已用于合成56种基于醌的化合物。他们针对引起锥虫病的寄生虫克氏锥虫进行了评估。此处描述的反应是该计划的一部分,该计划旨在利用现代,通用和高效的合成方法来一步一步制备锥虫杀菌化合物。我们已经鉴定出9种对寄生虫具有有效活性的化合物。其中3种药物的功效比苯甲硝唑(Bz)(用于治疗南美锥虫病的药物)高30倍。本文提供了涉及120多种化合物的反应的全面概述,旨在发现新的基于醌的骨架,该骨架具有抗T. cruzi的活性。。











































 京公网安备 11010802027423号
京公网安备 11010802027423号