当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Asymmetric N-alkylation of indoles with isatins catalyzed by N-heterocyclic carbene: efficient synthesis of functionalized cyclic N,O-aminal indole derivatives
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2020-05-26 , DOI: 10.1039/d0qo00237b Chengyuan Wang 1, 2, 3, 4, 5 , Zhuopeng Li 1, 2, 3, 4, 5 , Jiong Zhang 1, 2, 3, 4, 5 , Xin-Ping Hui 1, 2, 3, 4, 5
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2020-05-26 , DOI: 10.1039/d0qo00237b Chengyuan Wang 1, 2, 3, 4, 5 , Zhuopeng Li 1, 2, 3, 4, 5 , Jiong Zhang 1, 2, 3, 4, 5 , Xin-Ping Hui 1, 2, 3, 4, 5
Affiliation
|
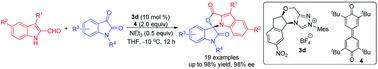
An N-heterocyclic carbene-catalyzed enantioselective N-alkylation of indole-2-formaldehydes with isatins has been achieved. Functionalized cyclic N,O-aminal indole derivatives were obtained in good yields with excellent enantioselectivities.
中文翻译:

N-杂环卡宾催化的吲哚与吲哚的不对称N-烷基化:功能化环状N,O-氨基吲哚衍生物的有效合成
已经实现了吲哚-2-甲醛与靛红的N-杂环卡宾催化的对映选择性N-烷基化。以良好的收率和优异的对映选择性获得了官能化的环状N,O-氨基吲哚衍生物。
更新日期:2020-06-30
中文翻译:

N-杂环卡宾催化的吲哚与吲哚的不对称N-烷基化:功能化环状N,O-氨基吲哚衍生物的有效合成
已经实现了吲哚-2-甲醛与靛红的N-杂环卡宾催化的对映选择性N-烷基化。以良好的收率和优异的对映选择性获得了官能化的环状N,O-氨基吲哚衍生物。









































 京公网安备 11010802027423号
京公网安备 11010802027423号