International Journal of Hydrogen Energy ( IF 8.1 ) Pub Date : 2020-05-25 , DOI: 10.1016/j.ijhydene.2020.04.237 Liping Cheng , Baoen Xu , Xiaoyan Li , Lingpeng Meng
|
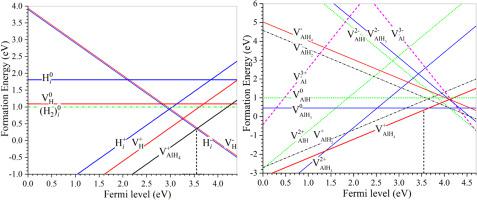
The roles of native defects and transition-metal additives (Ti, Sc and Ni) in the dehydrogenation of Mg(AlH4)2 hydride were investigated by First-principles calculations based on density functional theory. The elementary native defects including , , , , , and in Mg(AlH4)2 were identified. Based on the formation and migration of dominant defects, we proposed a dehydrogenation mechanism of Mg(AlH4)2. The formation of the dominant defect is the rate-limiting step in the dehydrogenation process. The highly mobile diffuses into the lattice and binds with Mg2+ to produce MgH2 phase. In the transition metal doped hydrides, interstitial defects Tii and Sci cause the Fermi-level shift to the left and lead to the decrease of the formation energy and activation energy of , which is beneficial to the dehydrogenation of Mg(AlH4)2. This study presents an in-depth understanding on the roles of native defects and transition metal additives in the dehydrogenation process of Mg(AlH4)2 hydride.
中文翻译:

天然缺陷和过渡金属添加剂在Mg(AlH 4)2脱氢机理中的作用
通过基于密度泛函理论的第一性原理计算,研究了天然缺陷和过渡金属添加剂(Ti,Sc和Ni)在Mg(AlH 4)2氢化物脱氢中的作用。基本的自然缺陷包括, , , , 和 在Mg(AlH 4)2中被鉴定。基于主要缺陷的形成和迁移,我们提出了Mg(AlH 4)2的脱氢机理。主要缺陷的形成是脱氢过程中的限速步骤。高度移动扩散到晶格中并与Mg 2+结合产生MgH 2相。在过渡金属掺杂的氢化物中,间隙缺陷Ti i和Sc i导致费米能级向左移动,并导致形成能和活化能的降低。,有利于Mg(AlH 4)2的脱氢。这项研究提出了对天然缺陷和过渡金属添加剂在Mg(AlH 4)2氢化物的脱氢过程中的作用的深入了解。











































 京公网安备 11010802027423号
京公网安备 11010802027423号