Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-05-24 , DOI: 10.1016/j.bmc.2020.115563 Paolo La Rocca 1 , Paola Rota 2 , Marco Piccoli 3 , Federica Cirillo 3 , Andrea Ghiroldi 3 , Valentina Franco 4 , Pietro Allevi 2 , Luigi Anastasia 5
|
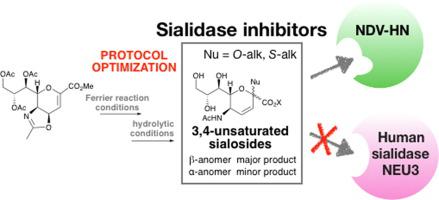
The optimization of the synthetic protocol to obtain the 3,4-unsaturated sialic acid derivatives, through the fine-tuning of both the Ferrier glycosylation conditions and the subsequent hydrolysis work-up, is herein reported. The accomplishment of the desired ® -anomers and some selected α-ones, in pure form, led us to evaluate their specific inhibitory activity towards NDV-HN and human sialidase NEU3. Importantly, the resulting data allowed the identification, for the first time, of three active 3,4 unsaturated sialic acid analogs, showing IC50 values against NDV-HN in the micromolar range.
中文翻译:

2β-3,4-不饱和唾液酸衍生物:作为纽卡斯尔病病毒血凝素神经氨酸酶抑制剂的合成优化和生物学评估。
本文报道了通过Ferrier糖基化条件和随后的水解后处理两者的微调来获得3,4-不饱和唾液酸衍生物的合成方案的优化。所需的-端基异构体和一些选定的α-原子的纯净形式,促使我们评估了它们对NDV-HN和人唾液酸酶NEU3的特异性抑制活性。重要的是,所得数据首次允许鉴定三种活性3,4不饱和唾液酸类似物,显示出在微摩尔范围内针对NDV-HN的IC 50值。











































 京公网安备 11010802027423号
京公网安备 11010802027423号