Molecular Cell ( IF 14.5 ) Pub Date : 2020-05-22 , DOI: 10.1016/j.molcel.2020.04.026 Muwen Kong 1 , Erin E Cutts 2 , Dongqing Pan 3 , Fabienne Beuron 2 , Thangavelu Kaliyappan 2 , Chaoyou Xue 1 , Edward P Morris 2 , Andrea Musacchio 3 , Alessandro Vannini 4 , Eric C Greene 1
|
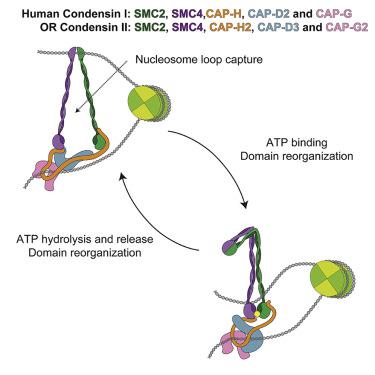
Structural maintenance of chromosomes (SMC) complexes are essential for genome organization from bacteria to humans, but their mechanisms of action remain poorly understood. Here, we characterize human SMC complexes condensin I and II and unveil the architecture of the human condensin II complex, revealing two putative DNA-entrapment sites. Using single-molecule imaging, we demonstrate that both condensin I and II exhibit ATP-dependent motor activity and promote extensive and reversible compaction of double-stranded DNA. Nucleosomes are incorporated into DNA loops during compaction without being displaced from the DNA, indicating that condensin complexes can readily act upon nucleosome-bound DNA molecules. These observations shed light on critical processes involved in genome organization in human cells.
中文翻译:

人类凝缩蛋白 I 和 II 驱动核小体结合 DNA 的广泛 ATP 依赖性压缩。
染色体结构维持 (SMC) 复合物对于从细菌到人类的基因组组织至关重要,但其作用机制仍知之甚少。在这里,我们表征了人类 SMC 复合物凝缩蛋白 I 和 II,并揭示了人类凝缩蛋白 II 复合物的结构,揭示了两个假定的 DNA 捕获位点。使用单分子成像,我们证明凝缩蛋白 I 和 II 都表现出 ATP 依赖性运动活性,并促进双链 DNA 的广泛和可逆压缩。核小体在压缩过程中并入 DNA 环中,而不会从 DNA 中移位,这表明凝缩蛋白复合物可以轻松地作用于核小体结合的 DNA 分子。这些观察结果揭示了人类细胞基因组组织中涉及的关键过程。











































 京公网安备 11010802027423号
京公网安备 11010802027423号