Journal of Fluorine Chemistry ( IF 1.7 ) Pub Date : 2020-05-06 , DOI: 10.1016/j.jfluchem.2020.109547 Vladimir O. Gelmboldt , Ivan O. Shyshkin , Vladimir Yu. Anisimov , Marina S. Fonari , Victor Ch. Kravtsov
|
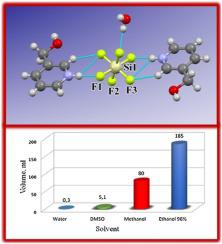
The hexafluorosilicate with the composition (LH)2SiF6·H2O (I, L = 3-hydroxymethylpyridine) was obtained by reaction of 45 % hexafluorosilicic acid with a methanol solution of L (H2SiF6 : L = 3: 1). The crystal structure I is stabilized by OH···O, NH···F, and OH···F H-bonds, and CH···F contacts. The symmetry of the SiF62– anion is close to D4h. The solubility of I in water was 1.63 mol. % and the degree of hydrolysis of the salt in a 1·10–4 M aqueous solution was 91.6 %. A PASS-analysis of the spectrum of biological activity L highlighted a low probability of toxic effects.
中文翻译:

双(3-羟甲基吡啶鎓)六氟硅酸盐一水合物作为新型潜在的抗龋剂:合成,晶体结构和药理性质
组成为(LH)2 SiF 6 ·H 2 O(I,L = 3-羟甲基吡啶)的六氟硅酸盐是通过使45%六氟硅酸与L的甲醇溶液(H 2 SiF 6:L = 3:1)反应获得的)。晶体结构I通过OH··O,NH···F和OH··F的H键稳定化,并且CH··F接触。SiF 6 2–阴离子的对称性接近D 4 h。I在水中的溶解度为1.63mol。%和盐的水解度为1·10 –4M水溶液为91.6%。对生物活性谱L的PASS分析强调了毒性作用的可能性较低。










































 京公网安备 11010802027423号
京公网安备 11010802027423号