当前位置:
X-MOL 学术
›
J. Fluorine Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Synthesis of N-substituted sulfonamides containing perhalopyridine moiety as bio-active candidates
Journal of Fluorine Chemistry ( IF 1.7 ) Pub Date : 2020-03-09 , DOI: 10.1016/j.jfluchem.2020.109507 Raziyeh Hosseini , Reza Ranjbar-Karimi , Kazem Mohammadiannejad
中文翻译:

含全卤吡啶部分作为生物活性候选物的N取代磺酰胺的合成
更新日期:2020-03-09
Journal of Fluorine Chemistry ( IF 1.7 ) Pub Date : 2020-03-09 , DOI: 10.1016/j.jfluchem.2020.109507 Raziyeh Hosseini , Reza Ranjbar-Karimi , Kazem Mohammadiannejad
|
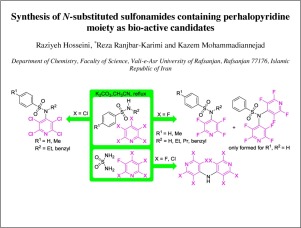
A series of new halogenated aryl sulfonamides, as bio-active candidates, was synthesized from the reaction of the corresponding aryl sulfonamides with pentafluoro- and pentachloropyridines. Surprisingly, unlike aryl sulfonamides, the reaction of sulfamides with pentafluoro- and pentachloropyridines gave unexpected bis-perfluoro(chloro)pyridin-4-ylamines.
中文翻译:

含全卤吡啶部分作为生物活性候选物的N取代磺酰胺的合成
通过相应的芳基磺酰胺与五氟和五氯吡啶的反应,合成了一系列新的卤化芳基磺酰胺作为生物活性候选物。令人惊讶地,与芳基磺酰胺不同,磺酰胺与五氟-和五氯吡啶的反应产生了出人意料的双全氟(氯)吡啶-4-基胺。











































 京公网安备 11010802027423号
京公网安备 11010802027423号