当前位置:
X-MOL 学术
›
Diam. Relat. Mater.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A novel electrochemical strategy for determination of vitamin B12 by Co(I/II) redox pair monitoring with boron-doped diamond electrode
Diamond and Related Materials ( IF 4.3 ) Pub Date : 2020-05-01 , DOI: 10.1016/j.diamond.2020.107793 Daniel F. Pereira , Edson R. Santana , Jamille V. Piovesan , Almir Spinelli
Diamond and Related Materials ( IF 4.3 ) Pub Date : 2020-05-01 , DOI: 10.1016/j.diamond.2020.107793 Daniel F. Pereira , Edson R. Santana , Jamille V. Piovesan , Almir Spinelli
|
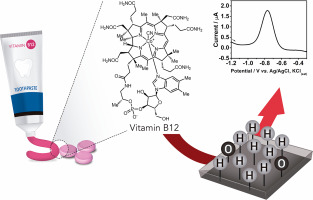
Abstract Vitamin B12 supplementation is recommended mainly for people who are vegetarian or vegan. The wide extent of the use of this supplementation should act as a warning sign regarding the quality of commercially available products. In this context, a novel electrochemical strategy is proposed for the determination of vitamin B12, where the Co(I/II) redox pair is monitored using a boron-doped diamond electrode for the analysis of supplementation products. The surface of the boron-doped diamond was characterized by electrochemical impedance spectroscopy and cyclic voltammetry. The electrical properties of the electrode are highly influenced by its surface terminations and a cathodic pretreatment of −2.0 V for 30 s in 0.5 mol L−1 H2SO4 before the analysis enhanced the analytical response. The cyclic voltammograms for vitamin B12 obtained at pH 5.0 showed four peaks: two oxidation peaks at −0.74 and +0.18 V, corresponding to Co(I/II) and Co(II/III) oxidations, respectively, and two reduction peaks at −0.12 and −0.75 V, corresponding to Co(III/II) and Co(II/I) reductions, respectively. The experimental parameters (pH, supporting electrolyte, pulse technique) were optimized for the monitoring of the Co(I/II) redox pair. The calibration plot for vitamin B12 was obtained by square wave voltammetry at pH 10.0. It was found to be linear from 0.25 to 5.0 μmol L−1, with a detection limit of 86.0 nmol L−1. A boron-doped diamond electrode with cathodic pretreatment was employed to determine vitamin B12 levels in fortified toothpaste and supplementation tablets. The results were compared with those provided by UV–vis spectrometry. The statistical analysis showed no significant difference between the two methodologies in terms of the precision and accuracy of the data obtained.
中文翻译:

一种通过掺杂硼金刚石电极监测 Co(I/II) 氧化还原对测定维生素 B12 的新型电化学策略
摘要 维生素 B12 补充剂主要推荐给素食者或纯素食者。这种补充剂的广泛使用应作为关于市售产品质量的警告标志。在此背景下,提出了一种用于测定维生素 B12 的新型电化学策略,其中使用掺硼金刚石电极监测 Co(I/II) 氧化还原对,以分析补充产品。掺硼金刚石的表面通过电化学阻抗谱和循环伏安法表征。在分析增强分析响应之前,电极的电性能受其表面终端和在 0.5 mol L-1 H2SO4 中 -2.0 V 阴极预处理 30 秒的高度影响。在 pH 5 下获得的维生素 B12 的循环伏安图。0 显示四个峰:-0.74 和 +0.18 V 处的两个氧化峰,分别对应于 Co(I/II) 和 Co(II/III) 氧化,以及 -0.12 和 -0.75 V 处的两个还原峰,对应于 Co (III/II) 和 Co(II/I) 减少,分别。优化了实验参数(pH、支持电解质、脉冲技术)以监测 Co(I/II) 氧化还原对。维生素 B12 的校准曲线是通过方波伏安法在 pH 10.0 下获得的。发现它在 0.25 到 5.0 μmol L-1 之间呈线性,检测限为 86.0 nmol L-1。使用经过阴极预处理的掺硼金刚石电极来测定强化牙膏和补充剂片剂中的维生素 B12 水平。将结果与紫外-可见光谱法提供的结果进行比较。
更新日期:2020-05-01
中文翻译:

一种通过掺杂硼金刚石电极监测 Co(I/II) 氧化还原对测定维生素 B12 的新型电化学策略
摘要 维生素 B12 补充剂主要推荐给素食者或纯素食者。这种补充剂的广泛使用应作为关于市售产品质量的警告标志。在此背景下,提出了一种用于测定维生素 B12 的新型电化学策略,其中使用掺硼金刚石电极监测 Co(I/II) 氧化还原对,以分析补充产品。掺硼金刚石的表面通过电化学阻抗谱和循环伏安法表征。在分析增强分析响应之前,电极的电性能受其表面终端和在 0.5 mol L-1 H2SO4 中 -2.0 V 阴极预处理 30 秒的高度影响。在 pH 5 下获得的维生素 B12 的循环伏安图。0 显示四个峰:-0.74 和 +0.18 V 处的两个氧化峰,分别对应于 Co(I/II) 和 Co(II/III) 氧化,以及 -0.12 和 -0.75 V 处的两个还原峰,对应于 Co (III/II) 和 Co(II/I) 减少,分别。优化了实验参数(pH、支持电解质、脉冲技术)以监测 Co(I/II) 氧化还原对。维生素 B12 的校准曲线是通过方波伏安法在 pH 10.0 下获得的。发现它在 0.25 到 5.0 μmol L-1 之间呈线性,检测限为 86.0 nmol L-1。使用经过阴极预处理的掺硼金刚石电极来测定强化牙膏和补充剂片剂中的维生素 B12 水平。将结果与紫外-可见光谱法提供的结果进行比较。











































 京公网安备 11010802027423号
京公网安备 11010802027423号