Tetrahedron ( IF 2.1 ) Pub Date : 2020-02-26 , DOI: 10.1016/j.tet.2020.131066 Shushuai Chen , Hongxin Liu , Wei Ye , Saini Li , Dongli Li , Zhaoming Liu , Weimin Zhang
|
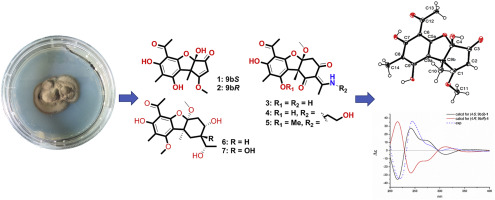
Ochuscins A‒G (1–7), seven highly oxygenated usnic acid derivatives were isolated from the deep-sea-derived fungus Ochroconis sp. FS449. Their structures were elucidated based on the 1D, 2D NMR, HRESIMS and single crystal X-ray diffraction, while the absolute configurations were established by the quantum chemical calculations of 13C NMR chemical shifts as well as ECD plots. The biosynthesis pathway of 1‒7 was proposed to generate via a dimerization of phloroglucinol derivatives. In the bioassay, compounds 2, 3, 4 and 7 exhibited moderate inhibitory activities against acetylcholinesterase with the IC50 values in the range from 49 to 80 μM. A preliminary structure-activity relationship was discussed.
中文翻译:

Ochuscins A‒G,来自深海真菌Ochroconis sp。的高度氧化的松萝酸衍生物。FS449
从深海来源的真菌Ochroconis sp。中分离出了Ochuscins A‒G(1 – 7),七个高度氧化的松萝酸衍生物。FS449。通过1D,2D NMR,HRESIMS和单晶X射线衍射阐明了它们的结构,同时通过13 C NMR化学位移的量子化学计算以及ECD图建立了绝对构型。的生物合成途径1 - 7提出了通过间苯三酚衍生物的二聚化以产生。在生物测定中,化合物2,3,4和7具有对乙酰胆碱酯酶的中等抑制活性,IC 50值为49至80μM。初步的构效关系进行了讨论。











































 京公网安备 11010802027423号
京公网安备 11010802027423号