当前位置:
X-MOL 学术
›
Chin. J. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Palladium(II)‐Catalyzed Aminotrifluoromethoxylation of Alkenes: Mechanistic Insight into the Effect of N‐Protecting Groups
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2020-02-18 , DOI: 10.1002/cjoc.201900516 Chaohuang Chen 1 , Chuanqi Hou 1 , Pinhong Chen 1 , Guosheng Liu 1, 2
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2020-02-18 , DOI: 10.1002/cjoc.201900516 Chaohuang Chen 1 , Chuanqi Hou 1 , Pinhong Chen 1 , Guosheng Liu 1, 2
Affiliation
|
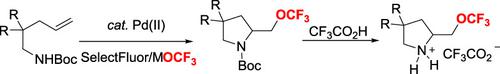
An efficient palladium‐catalyzed regioselective 5‐exo aminotrifluoromethoxylation of alkenes has been established herein, which provides a practical route towards the synthesis of OCF3‐containing pyrrolidines. tert‐Butyloxycarbonyl (Boc) as an amino protecting group plays a significant role in both the chemo‐ and regioselectivities. In addition, preliminary mechanistic studies reveal that the amino protecting group of substrates and the counter anion of palladium catalyst play critical roles in reaction efficiency presumably due to an isomerization of alkyl‐ Pd(II) intermediates. Moreover, the asymmetric 5‐exo aminotrifluoromethoxylation reaction has also been achieved by introducing a sterically bulky pyridinyl‐oxazoline ligand.
中文翻译:

钯(II)催化的烯烃氨基三氟甲氧基化:机理上对N保护基团作用的见解
本文已经建立了一种有效的钯催化烯烃的区域选择性5-外氨基三氟甲氧基化反应,为合成含OCF 3的吡咯烷提供了一条可行的途径。叔丁氧羰基(Boc)作为氨基保护基团在化学和区域选择性中都起着重要作用。此外,初步的机理研究表明,底物的氨基保护基团和钯催化剂的抗衡阴离子在反应效率中起着关键作用,大概是由于烷基-Pd(II)中间体的异构化。此外,还可以通过引入空间庞大的吡啶基-恶唑啉配体来实现不对称的5-exo氨基三氟甲氧基化反应。
更新日期:2020-02-18
中文翻译:

钯(II)催化的烯烃氨基三氟甲氧基化:机理上对N保护基团作用的见解
本文已经建立了一种有效的钯催化烯烃的区域选择性5-外氨基三氟甲氧基化反应,为合成含OCF 3的吡咯烷提供了一条可行的途径。叔丁氧羰基(Boc)作为氨基保护基团在化学和区域选择性中都起着重要作用。此外,初步的机理研究表明,底物的氨基保护基团和钯催化剂的抗衡阴离子在反应效率中起着关键作用,大概是由于烷基-Pd(II)中间体的异构化。此外,还可以通过引入空间庞大的吡啶基-恶唑啉配体来实现不对称的5-exo氨基三氟甲氧基化反应。










































 京公网安备 11010802027423号
京公网安备 11010802027423号