Acta Pharmaceutica Sinica B ( IF 14.7 ) Pub Date : 2020-01-27 , DOI: 10.1016/j.apsb.2020.01.013 Qinglan Guo 1 , Huan Xia 1 , Yuzhuo Wu 1 , Shuai Shao 1 , Chengbo Xu 1 , Tiantai Zhang 1 , Jiangong Shi 1
|
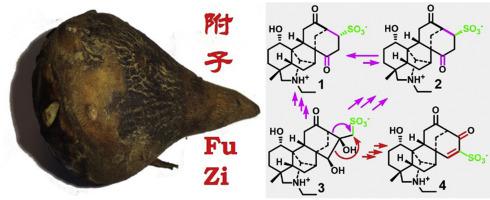
Three new C20-diterpenoid alkaloids with a sulfonic acid unit, named aconicarmisulfonines B and C (1 and 2) and chuanfusulfonine A (3), respectively, were isolated from the Aconitum carmichaelii lateral roots (“fu zi” in Chinese). Structures of 1–3 were determined by spectroscopic data analysis. Intriguing chemical properties and reactions were observed for the C20-diterpenoid alkaloids: (a) specific selective nucleophilic addition of the carbonyl (C-12) in 1 with CD3OD; (b) interconversion between 1 and 2 in D2O; (c) stereo- and/or regioselective deuterations of H-11α in 1–3 and both H-11α and H-11β in aconicarmisulfonine A (4); (d) TMSP-2,2,3,3-d4 promoted cleavage of the C-12−C-13 bond of 4 in D2O; (e) dehydrogenation of 4 in pyridine-d5, and (f) Na2SO3-assisted dehydrogenation and N-deethylation of songorine (5, a putative precursor of 1–4). Biogenetically, 1 and 2 are correlated with 4, for which the same novel carbon skeleton is proposed to be derived from semipinacol rearrangements via migrations of C-13−C-16 and C-15−C-16 bonds of the napelline-type skeleton, respectively. Meanwhile, 3 is a highly possible precursor or a concurrent product in the biosynthetic pathways of 1, 2, and 4. In the acetic acid-induced mice writhing assay, at 1.0 mg/kg (i.p.), compounds 1, 2, 5, 5a, and 5b exhibited analgesic effects against mice writhing.
中文翻译:

乌头中含有磺酸基的二萜生物碱的结构、性质、生物合成和活性
从乌头侧根中分离得到 3 个新的带有磺酸单元的 C 20 -二萜生物碱,分别命名为乌头磺胺 B 和 C ( 1和2 ) 以及川附磺宁 A ( 3 )。通过光谱数据分析确定1-3的结构。观察到 C 20 -二萜生物碱的有趣化学性质和反应:(a) 1中的羰基 (C-12) 与 CD 3 OD 发生特异性选择性亲核加成; (b)D 2 O中1和2之间的相互转化; (c) 1-3中 H-11 α以及乌头磺胺 A 中 H-11 α和 H-11 β的立体和/或区域选择性氘化 ( 4 ); (d) TMSP-2,2,3,3- d 4促进 D 2 O 中4的 C-12−C-13 键裂解; (e) 4在吡啶-d 5中脱氢,以及(f) Na 2 SO 3辅助松戈林脱氢和N-脱乙基化( 5 ,假定的1 – 4前体)。 在生物遗传学上, 1和2与4相关,其中相同的新型碳骨架被认为是通过napelline型骨架的C-13−C-16和C-15−C-16键的迁移从半频那醇重排衍生而来, 分别。同时, 3很可能是1、2和4生物合成途径中的前体或同时产物。在乙酸诱导的小鼠扭体测定中,在1.0mg /kg(ip )下,化合物1、2、5、5a和5b对小鼠扭体表现出镇痛作用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号