当前位置:
X-MOL 学术
›
Appl. Surf. Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Facile Preparation of Self-Assembled Ni/Co Phosphates Composite Spheres with Highly Efficient HER Electrocatalytic Performances
Applied Surface Science ( IF 6.3 ) Pub Date : 2020-04-01 , DOI: 10.1016/j.apsusc.2020.145383 Yuelong Xu , Ran Wang , Yuxuan Zheng , Lihui Zhang , Tifeng Jiao , Qiuming Peng , Zhenfa Liu
Applied Surface Science ( IF 6.3 ) Pub Date : 2020-04-01 , DOI: 10.1016/j.apsusc.2020.145383 Yuelong Xu , Ran Wang , Yuxuan Zheng , Lihui Zhang , Tifeng Jiao , Qiuming Peng , Zhenfa Liu
|
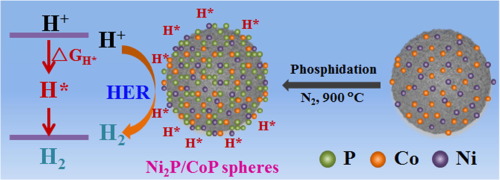
Abstract Because of their high catalytic activity and abundant resources, nonprecious transition metal phosphides are favorable substitutes for noble metals as hydrogen evolution reaction (HER) electrocatalysts. In this work, self-assembled Ni/Co phosphide composite N-doped carbon spheres were successfully prepared via a hydrothermal process, which had abundant exposed active sites for the HER. The as-obtained samples exhibited excellent electrocatalytic activity for the HER with an overpotential of only 160 mV (vs. RHE) at a current density of 10 mA cm−2 in a 0.5 M H2SO4 solution and a comparatively low Tafel slope (57 mV dec−1). Additionally, the Ni2P/CoP spheres also show good stability after 120 h and 5000 cycles of a cyclic voltammetry test. Their highly efficient catalytic activity for the HER is attributed to the specific spherical structure, which provides exposed Ni2P and CoP for favorable adsorption and desorption of hydrogen. Therefore, nonprecious transition metal phosphide spheres may provide a promising strategy to improve the catalytic activity of HER electrocatalysts.
中文翻译:

具有高效HER电催化性能的自组装Ni/Co磷酸盐复合球的简便制备
摘要 非贵金属过渡金属磷化物由于其催化活性高、资源丰富,是作为析氢反应(HER)电催化剂的贵金属的良好替代品。在这项工作中,通过水热法成功制备了自组装的 Ni/Co 磷化物复合 N 掺杂碳球,该球具有大量暴露的 HER 活性位点。所获得的样品对 HER 表现出优异的电催化活性,在 0.5 M H2SO4 溶液中的电流密度为 10 mA cm−2 和相对较低的 Tafel 斜率(57 mV dec -1). 此外,Ni2P/CoP 球体在循环伏安法测试 120 小时和 5000 次循环后也显示出良好的稳定性。它们对 HER 的高效催化活性归因于特定的球形结构,它提供了暴露的 Ni2P 和 CoP,有利于氢的吸附和解吸。因此,非贵重的过渡金属磷化物球可能为提高 HER 电催化剂的催化活性提供了一种有前景的策略。
更新日期:2020-04-01
中文翻译:

具有高效HER电催化性能的自组装Ni/Co磷酸盐复合球的简便制备
摘要 非贵金属过渡金属磷化物由于其催化活性高、资源丰富,是作为析氢反应(HER)电催化剂的贵金属的良好替代品。在这项工作中,通过水热法成功制备了自组装的 Ni/Co 磷化物复合 N 掺杂碳球,该球具有大量暴露的 HER 活性位点。所获得的样品对 HER 表现出优异的电催化活性,在 0.5 M H2SO4 溶液中的电流密度为 10 mA cm−2 和相对较低的 Tafel 斜率(57 mV dec -1). 此外,Ni2P/CoP 球体在循环伏安法测试 120 小时和 5000 次循环后也显示出良好的稳定性。它们对 HER 的高效催化活性归因于特定的球形结构,它提供了暴露的 Ni2P 和 CoP,有利于氢的吸附和解吸。因此,非贵重的过渡金属磷化物球可能为提高 HER 电催化剂的催化活性提供了一种有前景的策略。











































 京公网安备 11010802027423号
京公网安备 11010802027423号