当前位置:
X-MOL 学术
›
Appl. Surf. Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Carbon–based nanomaterials and ZnO ternary compound layers grown by laser technique for environmental and energy storage applications
Applied Surface Science ( IF 6.3 ) Pub Date : 2020-04-01 , DOI: 10.1016/j.apsusc.2020.145359 R. Ivan , C. Popescu , A. Pérez del Pino , C. Logofatu , E György
Applied Surface Science ( IF 6.3 ) Pub Date : 2020-04-01 , DOI: 10.1016/j.apsusc.2020.145359 R. Ivan , C. Popescu , A. Pérez del Pino , C. Logofatu , E György
|
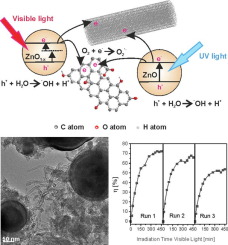
Abstract Carbon nanotube – highly reduced graphene oxide – transition metal oxide (ZnO) nanohybrid layers were synthesized using a one-step laser technique. Commercial multiwall carbon nanotubes (MWCNTs), graphene oxide (GO) platelets and ZnO nanoparticles were used as starting materials. We discuss the influence of carbon/metal oxide ratio on the physico-chemical properties of the nanohybrid layers, geometrical characteristics, shape and dimensions of constituent nanoentities, chemical composition and chemical bonding states, optical properties, UV–visible absorption, band gap values, as well as charge transfer properties. In the followings the relation between these properties and functional characteristics, removal of water contaminants, antibiotic molecules, and charge storage performances of the ternary, MWCNTs/reduced GO/ZnO layers are presented, identifying the optimum relative concentrations of the constituting nanomaterials. The high photocatalytic efficiencies both under UV and visible light irradiations, even after several consecutive degradation cycles, were attributed to effective separation of photogenerated charge carriers by carbon nanomaterials as well as formation of oxygen deficient ZnOx−1 nanocrystals. The enhanced charge storage capacity of ternary nanohybrid electrodes is based on combined electrochemical double layer capacitance and pseudocapacitance implying redox reactions on the surface and subsurface of the layers in contact with the electrolyte. Both functional properties are strongly influenced by the relative concentrations of the nanomaterials constituting the ternary layers.
中文翻译:

用于环境和储能应用的激光技术生长的碳基纳米材料和 ZnO 三元化合物层
摘要 碳纳米管 - 高度还原的氧化石墨烯 - 过渡金属氧化物 (ZnO) 纳米混合层是使用一步激光技术合成的。商业多壁碳纳米管 (MWCNT)、氧化石墨烯 (GO) 薄片和 ZnO 纳米颗粒被用作起始材料。我们讨论了碳/金属氧化物比例对纳米杂化层的物理化学性质、几何特征、组成纳米实体的形状和尺寸、化学成分和化学键合状态、光学性质、紫外-可见吸收、带隙值、以及电荷转移特性。以下是这些性质与功能特性、去除水污染物、抗生素分子和三元电荷存储性能之间的关系,呈现了 MWCNTs/还原的 GO/ZnO 层,确定了构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。确定构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。确定构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。
更新日期:2020-04-01
中文翻译:

用于环境和储能应用的激光技术生长的碳基纳米材料和 ZnO 三元化合物层
摘要 碳纳米管 - 高度还原的氧化石墨烯 - 过渡金属氧化物 (ZnO) 纳米混合层是使用一步激光技术合成的。商业多壁碳纳米管 (MWCNT)、氧化石墨烯 (GO) 薄片和 ZnO 纳米颗粒被用作起始材料。我们讨论了碳/金属氧化物比例对纳米杂化层的物理化学性质、几何特征、组成纳米实体的形状和尺寸、化学成分和化学键合状态、光学性质、紫外-可见吸收、带隙值、以及电荷转移特性。以下是这些性质与功能特性、去除水污染物、抗生素分子和三元电荷存储性能之间的关系,呈现了 MWCNTs/还原的 GO/ZnO 层,确定了构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。确定构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。确定构成纳米材料的最佳相对浓度。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。在紫外线和可见光照射下的高光催化效率,即使经过几个连续的降解循环,归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。归因于碳纳米材料对光生载流子的有效分离以及缺氧 ZnOx-1 纳米晶体的形成。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。三元纳米混合电极增强的电荷存储能力基于电化学双层电容和赝电容的组合,这意味着与电解质接触的层的表面和亚表面发生氧化还原反应。这两种功能特性都受到构成三元层的纳米材料的相对浓度的强烈影响。











































 京公网安备 11010802027423号
京公网安备 11010802027423号