当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Stereoselective Synthesis of β-Amino Ynones by the Addition of Alkynones to Nonracemic Sulfinimines: Formal Total Synthesis of l-Xylo and l-Arabino Phytosphingosines.
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-01-16 , DOI: 10.1021/acs.joc.9b02938 Polimera Obula Reddy 1 , Arava Amaranadha Reddy 1 , Kavirayani R Prasad 1
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-01-16 , DOI: 10.1021/acs.joc.9b02938 Polimera Obula Reddy 1 , Arava Amaranadha Reddy 1 , Kavirayani R Prasad 1
Affiliation
|
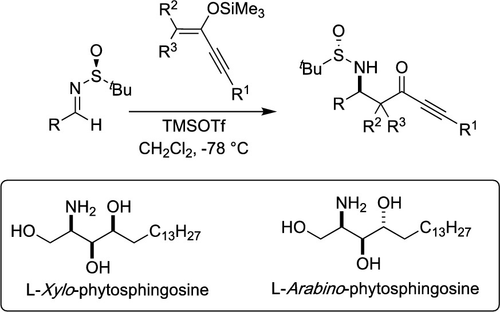
The addition of silyl enol ethers obtained from ynones to sulfinimines furnished the corresponding β-sulfinamido ynones in a very good yield and diastereoselectivity. The formed ynones serve as precursors amenable for the synthesis of bioactive compounds. This has been illustrated in the synthesis of l-xylo and l-arabino phytosphingosines.
中文翻译:

通过将炔烃加到非外消旋亚磺胺上的立体选择性合成β-氨基壬烯:l-木糖和l-阿拉伯植物鞘氨醇的正式全合成。
将由炔酮得到的甲硅烷基烯醇醚加到亚磺胺中,以非常好的收率和非对映选择性提供了相应的β-亚磺酰胺基炔酮。形成的炔酮用作适于合成生物活性化合物的前体。在1-xylo和1-阿拉伯糖基植物鞘氨醇的合成中已经说明了这一点。
更新日期:2020-01-17
中文翻译:

通过将炔烃加到非外消旋亚磺胺上的立体选择性合成β-氨基壬烯:l-木糖和l-阿拉伯植物鞘氨醇的正式全合成。
将由炔酮得到的甲硅烷基烯醇醚加到亚磺胺中,以非常好的收率和非对映选择性提供了相应的β-亚磺酰胺基炔酮。形成的炔酮用作适于合成生物活性化合物的前体。在1-xylo和1-阿拉伯糖基植物鞘氨醇的合成中已经说明了这一点。











































 京公网安备 11010802027423号
京公网安备 11010802027423号