当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Nickel/NHC‐Catalyzed Enantioselective Cyclization of Pyridones and Pyrimidones with Tethered Alkenes
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-01-22 , DOI: 10.1002/adsc.201901582 Di Shen 1, 2 , Wu‐Bin Zhang 2 , Zhiyang Li 2 , Shi‐Liang Shi 2 , Youjun Xu 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-01-22 , DOI: 10.1002/adsc.201901582 Di Shen 1, 2 , Wu‐Bin Zhang 2 , Zhiyang Li 2 , Shi‐Liang Shi 2 , Youjun Xu 1
Affiliation
|
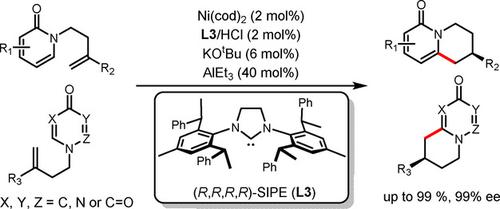
Reported is a highly enantioselective Ni(0)‐catalyzed endo‐selective C−H annulation of 2‐ and 4‐pyridone, and 4‐pyrimidone with alkenes to provide drug‐relevant bicyclic heterocycle products. The use of a readily prepared chiral bulky NHC ligand (SIPE) for Ni catalyst and commercially available AlEt3 as co‐catalyst enhanced the practicality of this reaction.
中文翻译:

镍/ NHC催化的吡啶酮和嘧啶与链状烯烃的对映选择性环化
报道了2-和4-吡啶酮和4-嘧啶酮与烯烃的高度对映选择性Ni(0)催化的内选择性CH环化反应,可提供与药物相关的双环杂环产物。使用易于制备的手性庞大的NHC配体(SIPE)作为Ni催化剂和市售的AlEt 3作为助催化剂,可以提高该反应的实用性。
更新日期:2020-01-23
中文翻译:

镍/ NHC催化的吡啶酮和嘧啶与链状烯烃的对映选择性环化
报道了2-和4-吡啶酮和4-嘧啶酮与烯烃的高度对映选择性Ni(0)催化的内选择性CH环化反应,可提供与药物相关的双环杂环产物。使用易于制备的手性庞大的NHC配体(SIPE)作为Ni催化剂和市售的AlEt 3作为助催化剂,可以提高该反应的实用性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号