Bioorganic & Medicinal Chemistry Letters ( IF 2.7 ) Pub Date : 2018-11-09 , DOI: 10.1016/j.bmcl.2018.11.018 Anton Shetnev , Angelina Osipyan , Sergey Baykov , Alexander Sapegin , Zhanna Chirkova , Michail Korsakov , Anél Petzer , Idalet Engelbrecht , Jacobus P. Petzer
|
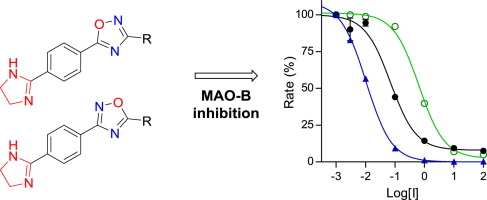
Series of structurally diverse 2-imidazoline derivatives have been synthesized by condensation of substituted aldehydes with ethylenediamine, Pd-catalyzed N-arylation of 2-imidazolines and by the formation of 1,2,4-oxadiazoles and benzoxazepines from 2-imidazoline-containing precursors. The 2-imidazoline derivatives were evaluated as potential inhibitors of human monoamine oxidase (MAO) A and B. Among the 2-imidazolines, good potency inhibitors were discovered with compound 9p (IC50 = 0.012 µM) being the most potent MAO-B inhibitor, while compound 9d (IC50 = 0.751 µM) was the most potent MAO-A inhibitor of the series. These potencies are in the same range as those of reference MAO inhibitors used in the clinic. Among 33 compounds evaluated, 13 exhibited IC50 values in the submicromolar range for the inhibition of an MAO isoform. It is postulated that the imidazoline moieties of some of these inhibitors may be recognized by the imidazoline I2-binding site of MAO. Good potency MAO inhibitors may be useful for the treatment of neuropsychiatric and neurodegenerative disorders such as depression and Parkinson’s disease, and future application for the treatment of prostate cancer, congestive heart failure and Alzheimer’s disease. In addition, high potency 2-imidazoline-derived MAO inhibitors may be used as potential probes for the imidazoline binding sites of the MAOs, as well as to determine alternative binding regions of imidazoline within the MAO active site.
中文翻译:

基于特权的2-咪唑啉分子框架的新型单胺氧化酶抑制剂
通过取代的醛与乙二胺的缩合,Pd催化的2-咪唑啉的N-芳基化反应以及由含2-咪唑啉的前体形成1,2,4-恶二唑和苯并氮杂品的合成,合成了一系列结构多样的2-咪唑啉衍生物。 。评价了2-咪唑啉衍生物作为人单胺氧化酶(MAO)A和B的潜在抑制剂。在2-咪唑啉中,发现了有效的抑制剂,其中化合物9p(IC 50 = 0.012 µM)是最有效的MAO-B抑制剂。 ,而化合物9d(IC 50 = 0.751 µM)是该系列中最有效的MAO-A抑制剂。这些效力与临床中使用的参考MAO抑制剂的效力相同。在评估的33种化合物中,有13种显示出在亚微摩尔范围内的IC 50值,可抑制MAO同种型。据推测,这些抑制剂中的一些的咪唑啉部分可以被咪唑啉I 2识别。-MAO的结合位点。高效的MAO抑制剂可用于治疗神经精神疾病和神经退行性疾病,例如抑郁症和帕金森氏病,以及将来在治疗前列腺癌,充血性心力衰竭和阿尔茨海默氏病中的应用。另外,高效2-咪唑啉衍生的MAO抑制剂可以用作MAO的咪唑啉结合位点的潜在探针,以及确定MAO活性位点内咪唑啉的替代结合区。



























 京公网安备 11010802027423号
京公网安备 11010802027423号