Analytical and Bioanalytical Chemistry ( IF 3.8 ) Pub Date : 2018-11-06 , DOI: 10.1007/s00216-018-1457-0 Philippe Corbisier , Hendrik Emons
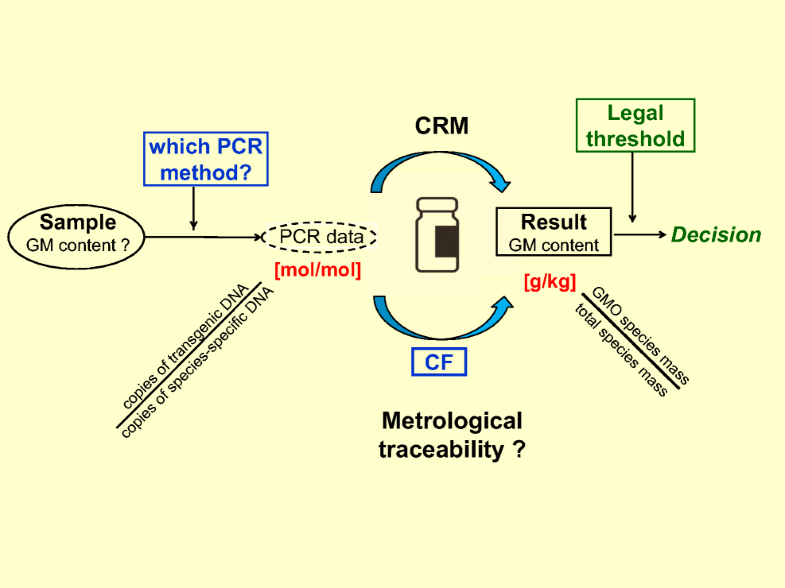
The GM content in a food or feed product produced from or containing genetically modified organisms (GMO) has to be expressed in Europe in the form of a GM mass fraction. However, the most widely used quantification methods, based on PCR, are basically counting PCR-amplifiable DNA fragments in a sample extract. This paper outlines the requirements for obtaining comparable measurement results which are fit for regulatory decision-making. It introduces the concept of a reference measurement system which enables GMO analysis laboratories to relate their results to a universally accessible reference, thus establishing metrological traceability to a unique reference point. The conversion factors required for transforming data from one measurement unit into the other have to carry a minimum uncertainty and are anchored to specified certified reference materials. The establishment of such conversion factors and related calibration approaches to achieve comparable GM quantification results are sketched.
ᅟ
中文翻译:

迈向GM量化的可溯源和可比较的结果
在欧洲,由转基因生物(GMO)生产或含有转基因生物(GMO)的食品或饲料中的转基因含量必须以转基因质量分数的形式表示。但是,基于PCR的最广泛使用的定量方法基本上是对样品提取物中可PCR扩增的DNA片段进行计数。本文概述了获得适合监管决策的可比较测量结果的要求。它介绍了参考测量系统的概念,该系统使GMO分析实验室可以将其结果与通用参考相关联,从而建立了到唯一参考点的计量溯源性。将数据从一个测量单位转换为另一个测量单位所需的转换系数必须具有最小的不确定性,并固定在指定的经过认证的参考材料上。概述了建立此类转换因子的方法和相关的校准方法,以实现可比的GM定量结果。

ᅟ











































 京公网安备 11010802027423号
京公网安备 11010802027423号