当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Chemoselective Pd‐Catalyzed C‐TeCF3 Coupling of Aryl Iodides
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2018-11-20 , DOI: 10.1002/anie.201810950 Theresa Sperger 1 , Sinem Guven 1 , Franziska Schoenebeck 1
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2018-11-20 , DOI: 10.1002/anie.201810950 Theresa Sperger 1 , Sinem Guven 1 , Franziska Schoenebeck 1
Affiliation
|
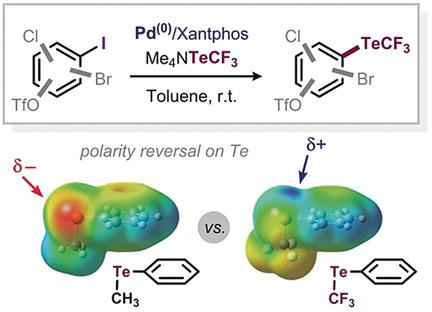
While the TeCF3 moiety features promising properties and potential in a range of applications, no direct synthetic method exists for its incorporation into aromatic scaffolds. This report features the first direct catalytic method for the formation of C(sp2)−TeCF3 bonds. The method relies on a Pd/Xantphos catalytic system and allows for the trifluoromethyltellurolation of aryl iodides. Our computational and experimental mechanistic analyses shed light on the privileged activity of Xantphos in this transformation.
中文翻译:

芳基碘化物的化学选择性Pd催化C-TeCF3偶联
尽管TeCF 3部分在一系列应用中具有令人鼓舞的特性和潜力,但尚无直接合成方法将其掺入芳族支架中。该报告的特点是形成C(sp 2)-TeCF 3键的第一种直接催化方法。该方法依赖于Pd / Xantphos催化体系,并允许芳基碘化物的三氟甲基碲酸化。我们的计算和实验机制分析揭示了Xantphos在此转换中的特权活动。
更新日期:2018-11-20
中文翻译:

芳基碘化物的化学选择性Pd催化C-TeCF3偶联
尽管TeCF 3部分在一系列应用中具有令人鼓舞的特性和潜力,但尚无直接合成方法将其掺入芳族支架中。该报告的特点是形成C(sp 2)-TeCF 3键的第一种直接催化方法。该方法依赖于Pd / Xantphos催化体系,并允许芳基碘化物的三氟甲基碲酸化。我们的计算和实验机制分析揭示了Xantphos在此转换中的特权活动。









































 京公网安备 11010802027423号
京公网安备 11010802027423号