当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Multi-component synthesis of 3-substituted indoles and their cyclisation to α-carbolines via I2-promoted intramolecular C2 oxidative amination/aromatisation at room temperature
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2018-10-17 , DOI: 10.1039/c8ob02362j Bhaskar Deka 1, 2, 3, 4, 5 , Pranjal K. Baruah 1, 2, 3, 4, 5 , Mohit L. Deb 1, 2, 3, 4, 5
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2018-10-17 , DOI: 10.1039/c8ob02362j Bhaskar Deka 1, 2, 3, 4, 5 , Pranjal K. Baruah 1, 2, 3, 4, 5 , Mohit L. Deb 1, 2, 3, 4, 5
Affiliation
|
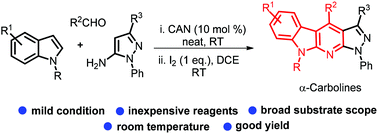
Condensation of indoles, aldehydes and pyrazol-5-amine in the presence of ceric ammonium nitrate gives 3-substituted indoles. These then cyclise to α-carbolines at room temperature through I2-promoted intramolecular C2 amination and aromatisation in open air. A plausible mechanism is proposed based on some controlled experiments.
中文翻译:

3-取代的吲哚的多组分合成及其在室温下通过I 2促进的分子内C2氧化胺化/芳构化将其环化为α-咔啉
在硝酸铈铵存在下,吲哚,醛和吡唑-5-胺的缩合得到3-取代的吲哚。然后它们在室温下通过I 2促进的分子内C2胺化和在露天的芳构化作用环化成α-咔啉。基于一些受控实验,提出了一种合理的机制。
更新日期:2018-11-02
中文翻译:

3-取代的吲哚的多组分合成及其在室温下通过I 2促进的分子内C2氧化胺化/芳构化将其环化为α-咔啉
在硝酸铈铵存在下,吲哚,醛和吡唑-5-胺的缩合得到3-取代的吲哚。然后它们在室温下通过I 2促进的分子内C2胺化和在露天的芳构化作用环化成α-咔啉。基于一些受控实验,提出了一种合理的机制。











































 京公网安备 11010802027423号
京公网安备 11010802027423号