当前位置:
X-MOL 学术
›
Chem. Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Regio- and chemoselective Csp3–H arylation of benzylamines by single electron transfer/hydrogen atom transfer synergistic catalysis†
Chemical Science ( IF 7.6 ) Pub Date : 2018-09-12 00:00:00 , DOI: 10.1039/c8sc02965b Takafumi Ide 1 , Joshua P Barham 1 , Masashi Fujita 1 , Yuji Kawato 1 , Hiromichi Egami 1 , Yoshitaka Hamashima 1
Chemical Science ( IF 7.6 ) Pub Date : 2018-09-12 00:00:00 , DOI: 10.1039/c8sc02965b Takafumi Ide 1 , Joshua P Barham 1 , Masashi Fujita 1 , Yuji Kawato 1 , Hiromichi Egami 1 , Yoshitaka Hamashima 1
Affiliation
|
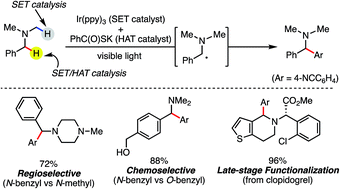
We present a highly regio- and chemoselective Csp3–H arylation of benzylamines mediated by synergy of single electron transfer (SET) and hydrogen atom transfer (HAT) catalysis. Under well precedented SET catalysis alone, the arylation reaction of N,N-dimethylbenzylamine proceeded via aminium radical cation formation and selectively targeted the N-methyl group. In contrast, addition of PhC(O)SH as a HAT catalyst precursor completely switched the regioselectivity to Csp3–H arylation at the N-benzylic position. Measurement of oxidation potentials indicated that the conjugate base of PhC(O)SH is oxidized in preference to the substrate amine. The discovery of the thiocarboxylate as a novel HAT catalyst allowed for the selective generation of the sulfur-centered radical, so that the N-benzyl selectivity was achieved by overriding the inherent N-methyl and/or N-methylene selectivity under SET catalysis conditions. While visible light-driven α-C–H functionalization of amines has mostly been demonstrated with aniline derivatives and tetrahydroisoquinolines (THIQs), our method is applicable to a variety of primary, secondary and tertiary benzylamines for efficient N-benzylic C–H arylation. Functional group tolerance was high, and various 1,1-diarylmethylamines, including an α,α,α-trisubstituted amine, were obtained in good to excellent yield (up to 98%). Importantly, the reaction is applicable to late-stage functionalization of pharmaceuticals.
中文翻译:

通过单电子转移/氢原子转移协同催化对苯甲胺进行区域和化学选择性 Csp3–H 芳基化†
我们提出了通过单电子转移(SET)和氢原子转移(HAT)催化的协同作用介导的苄胺的高度区域和化学选择性Csp 3 -H芳基化。仅在先例的 SET 催化下, N , N-二甲基苄胺的芳基化反应通过铵自由基阳离子的形成进行,并选择性地靶向N-甲基。相比之下,添加PhC(O)SH作为HAT催化剂前体将区域选择性完全转变为N-苄基位置处的Csp 3 –H芳基化。氧化电位的测量表明,PhC(O)SH 的共轭碱优先于底物胺被氧化。硫代羧酸酯作为新型 HAT 催化剂的发现允许选择性生成以硫为中心的自由基,从而通过在 SET 催化条件下超越固有的N-甲基和/或N-亚甲基选择性来实现N-苄基选择性。虽然可见光驱动的胺 α-C-H 官能化主要是用苯胺衍生物和四氢异喹啉 (THIQ) 来证明的,但我们的方法适用于各种伯、仲和叔苄胺,以实现有效的N-苄基 C-H 芳基化。官能团耐受性高,各种 1,1-二芳基甲胺(包括 α,α,α-三取代胺)的产率良好至优异(高达 98%)。重要的是,该反应适用于药物的后期功能化。
更新日期:2018-09-12
中文翻译:

通过单电子转移/氢原子转移协同催化对苯甲胺进行区域和化学选择性 Csp3–H 芳基化†
我们提出了通过单电子转移(SET)和氢原子转移(HAT)催化的协同作用介导的苄胺的高度区域和化学选择性Csp 3 -H芳基化。仅在先例的 SET 催化下, N , N-二甲基苄胺的芳基化反应通过铵自由基阳离子的形成进行,并选择性地靶向N-甲基。相比之下,添加PhC(O)SH作为HAT催化剂前体将区域选择性完全转变为N-苄基位置处的Csp 3 –H芳基化。氧化电位的测量表明,PhC(O)SH 的共轭碱优先于底物胺被氧化。硫代羧酸酯作为新型 HAT 催化剂的发现允许选择性生成以硫为中心的自由基,从而通过在 SET 催化条件下超越固有的N-甲基和/或N-亚甲基选择性来实现N-苄基选择性。虽然可见光驱动的胺 α-C-H 官能化主要是用苯胺衍生物和四氢异喹啉 (THIQ) 来证明的,但我们的方法适用于各种伯、仲和叔苄胺,以实现有效的N-苄基 C-H 芳基化。官能团耐受性高,各种 1,1-二芳基甲胺(包括 α,α,α-三取代胺)的产率良好至优异(高达 98%)。重要的是,该反应适用于药物的后期功能化。











































 京公网安备 11010802027423号
京公网安备 11010802027423号