Bioorganic Chemistry ( IF 4.5 ) Pub Date : 2018-09-06 , DOI: 10.1016/j.bioorg.2018.08.040 Lingnan Li , Miaomiao Zhou , Guimin Xue , Wenli Wang , Xuwei Zhou , Xiaobing Wang , Lingyi Kong , Jianguang Luo
|
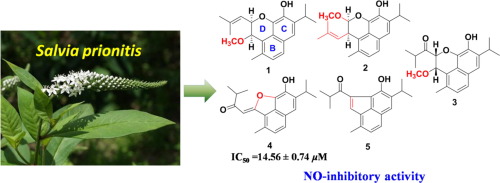
Five previously undescribed 4,5-seco-abietane rearrange diterpenoids (1–5, Prionidipene A-E) were isolated from the aerial parts of Salvia prionitis, along with thirteen known seco-abietane diterpenoids (6–18). The structures of 1–5 were elucidated mainly based on analysis of NMR and MS data. The absolute configurations of 1–3 were determined by evaluation of experimental and calculated electronic circular dichroism (ECD) spectra. Putative biosynthetic pathways toward the formation of 1 and 2 are proposed. The nitric oxide (NO) production inhibitory effects of all isolates in lipopolysaccharide (LPS)-induced in RAW 264.7 cells were evaluated. Interestingly, compounds 4 and 9 with a furan-ring showed potent inhibitory activity with IC50 values of 14.56 and 15.11 μM, respectively.
中文翻译:

生物活性塞科从地上部分-abietane重排二萜红根草
五个以前未描述-4,5-开环-abietane重排二萜类化合物(1 - 5, Prionidipene AE)从地上部分分离红根草,用13个沿着已知山高-abietane二萜类化合物(6 - 18)。主要根据NMR和MS数据分析来阐明1 – 5的结构。的绝对构型1 - 3通过实验的评估和计算电子圆二色性(ECD)光谱确定。推定的1和2形成的生物合成途径被提议。评估了所有分离株在RAW 264.7细胞中诱导的脂多糖(LPS)中一氧化氮(NO)产生的抑制作用。有趣的是,具有呋喃环的化合物4和9表现出强大的抑制活性,IC 50值分别为14.56和15.11μM。











































 京公网安备 11010802027423号
京公网安备 11010802027423号