Catalysis Communications ( IF 3.4 ) Pub Date : 2018-08-12 , DOI: 10.1016/j.catcom.2018.08.007 Marcelo Vilches-Herrera , Matias Concha-Puelles , Nicole Carvajal , Juana Molina , Rocio Santander , Marcos Caroli Rezende , Susan Lühr
|
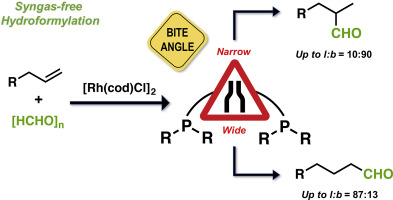
The correlation between the activity, regio- and chemoselectivity of Rh-diphosphine catalyst and the ligand bite natural angle (βn) in the syngas-free hydroformylation of allyl cyanide was investigated. A screening of Xantphos type and diphosphine alkyl ligands with different bite natural angles was studied. Interesting, a switch in the linear to the branch regioselectivity was found. Wide βn favour a linear regioselectivity whereas smaller βn allow the formation of the branched aldehyde as the major product. Modification of the substituents at the phosphorus atoms of the diphosphine ligands produced a dramatic change in the hydroformylation. Others β-functionalized olefins were also branched hydroformylated.
中文翻译:

齿状二膦配体的咬合自然角在β-官能化烯烃的无合成气支链羰基化反应中的影响
活性,区域选择性和化学选择性的Rh-二膦催化剂和配位体咬合自然角度(β之间的相关性Ñ在烯丙基腈的自由合成气-加氢甲酰化)进行了研究。研究了不同咬合自然角度的Xantphos类型和二膦烷基配体的筛选。有趣的是,发现了线性到分支区域选择性的切换。宽β Ñ青睐的线性区域选择性而较小的β Ñ允许的支链醛作为主要产物的形成。在二膦配体的磷原子上的取代基的修饰在加氢甲酰化中产生了显着变化。其他β-官能化的烯烃也被支链加氢甲酰化。









































 京公网安备 11010802027423号
京公网安备 11010802027423号