当前位置:
X-MOL 学术
›
Chem. Eur. J.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Decarboxylative Organocatalytic Allylic Amination of Morita–Baylis–Hillman Carbamates
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2018-08-17 , DOI: 10.1002/chem.201803677 Vojtěch Dočekal 1 , Michal Šimek 1 , Martin Dračínský 2 , Jan Veselý 1
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2018-08-17 , DOI: 10.1002/chem.201803677 Vojtěch Dočekal 1 , Michal Šimek 1 , Martin Dračínský 2 , Jan Veselý 1
Affiliation
|
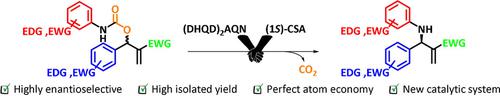
The present study reports the organocatalytic enantioselective allylic amination of Morita–Baylis–Hillman carbamates efficiently catalyzed by a chiral amine in the presence of a Brønsted acid. Chiral allylic amines were produced in high yields (up to 98 %) and enantioselectivities (up to 97 % ee). This method provides an efficient and easily performed route to prepare α‐methylene‐β‐lactams, and other optically active β‐lactams, such as the cholesterol‐lowering drug Ezetimibe.
中文翻译:

Morita–Baylis–Hillman氨基甲酸酯的脱羧有机催化烯丙基胺化
本研究报道了在布朗斯台德酸存在下,手性胺能有效催化森田-贝利斯-希尔曼氨基甲酸酯的有机催化对映选择性烯丙基胺化。手性烯丙基胺的收率高(高达98%)和对映选择性(高达97%ee)。该方法为制备α-亚甲基-β-内酰胺和其他旋光性β-内酰胺(例如降低胆固醇的药物Ezetimibe)提供了有效且容易执行的途径。
更新日期:2018-08-17
中文翻译:

Morita–Baylis–Hillman氨基甲酸酯的脱羧有机催化烯丙基胺化
本研究报道了在布朗斯台德酸存在下,手性胺能有效催化森田-贝利斯-希尔曼氨基甲酸酯的有机催化对映选择性烯丙基胺化。手性烯丙基胺的收率高(高达98%)和对映选择性(高达97%ee)。该方法为制备α-亚甲基-β-内酰胺和其他旋光性β-内酰胺(例如降低胆固醇的药物Ezetimibe)提供了有效且容易执行的途径。











































 京公网安备 11010802027423号
京公网安备 11010802027423号