当前位置:
X-MOL 学术
›
Eur. J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
8‐exo‐dig‐Selective Cycloisomerization for the Synthesis of Dibenzo[b,e][1,4]diazocines Using Cationic AuI Catalysts
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2018-08-23 , DOI: 10.1002/ejoc.201801037 Mamoru Ito 1 , Daisuke Inoue 1 , Asahi Takaki 1 , Kyalo Stephen Kanyiva 2 , Takanori Shibata 1
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2018-08-23 , DOI: 10.1002/ejoc.201801037 Mamoru Ito 1 , Daisuke Inoue 1 , Asahi Takaki 1 , Kyalo Stephen Kanyiva 2 , Takanori Shibata 1
Affiliation
|
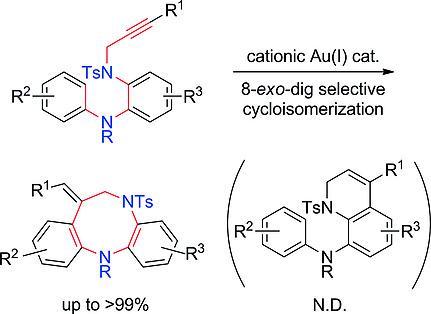
The cationic AuI‐catalyzed intramolecular reaction of N‐propargyl‐2‐anilinoanilines gave a diazocine skeleton via 8‐exo‐dig‐selective cycloisomerization. Both terminal and internal alkynes could be used according to the choice of ligand of the AuI complex. Two nitrogen atoms in the tether of substrates were critical in this selective transformation.

中文翻译:

阳离子AuI催化剂的8-exo-dig-选择性环异构化反应合成二苯并[b,e] [1,4]重氮化合物
所述阳离子金我催化的分子内反应Ñ -propargyl -2- anilinoanilines给予了二氮芳辛骨架经由8-外型-挖-选择性环异构。根据Au I配合物的配体选择,可以使用末端炔烃和内部炔烃。底物的系链中的两个氮原子对于这种选择性转化至关重要。

更新日期:2018-08-23

中文翻译:

阳离子AuI催化剂的8-exo-dig-选择性环异构化反应合成二苯并[b,e] [1,4]重氮化合物
所述阳离子金我催化的分子内反应Ñ -propargyl -2- anilinoanilines给予了二氮芳辛骨架经由8-外型-挖-选择性环异构。根据Au I配合物的配体选择,可以使用末端炔烃和内部炔烃。底物的系链中的两个氮原子对于这种选择性转化至关重要。












































 京公网安备 11010802027423号
京公网安备 11010802027423号