当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Tetra-n-butylammonium bromide (TBAB)-initiated carbonylation–peroxidation of styrene derivatives with aldehydes and hydroperoxides†
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2018-07-16 00:00:00 , DOI: 10.1039/c8qo00525g Yao Yao 1, 2, 3, 4, 5 , Zhen Wang 1, 2, 3, 4, 5 , Bin Wang 1, 2, 3, 4, 5
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2018-07-16 00:00:00 , DOI: 10.1039/c8qo00525g Yao Yao 1, 2, 3, 4, 5 , Zhen Wang 1, 2, 3, 4, 5 , Bin Wang 1, 2, 3, 4, 5
Affiliation
|
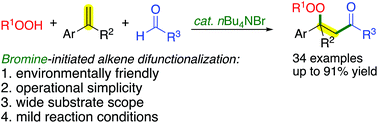
Tetra-n-butylammonium bromide (TBAB)-catalyzed carbonylation–peroxidation of styrene derivatives is described, which allows for the synthesis of β-peroxy ketones under mild conditions. The ease of handling, low cost, and environmentally friendly bromine catalyst made this protocol very practical. Mechanistic studies revealed that it was a bromine initiated radical difunctionalization of alkenes.
中文翻译:

四正丁基溴化铵(TBAB)引发的苯乙烯衍生物与醛和氢过氧化物的羰基化-过氧化反应†
描述了四正丁基溴化铵(TBAB)催化的苯乙烯衍生物的羰基化-过氧化,可在温和条件下合成β-过氧酮。易于操作,低成本和环保的溴催化剂使该协议非常实用。机理研究表明,这是溴引发的烯烃自由基双官能化。
更新日期:2018-07-16
中文翻译:

四正丁基溴化铵(TBAB)引发的苯乙烯衍生物与醛和氢过氧化物的羰基化-过氧化反应†
描述了四正丁基溴化铵(TBAB)催化的苯乙烯衍生物的羰基化-过氧化,可在温和条件下合成β-过氧酮。易于操作,低成本和环保的溴催化剂使该协议非常实用。机理研究表明,这是溴引发的烯烃自由基双官能化。











































 京公网安备 11010802027423号
京公网安备 11010802027423号