Analytical and Bioanalytical Chemistry ( IF 3.8 ) Pub Date : 2018-04-28 , DOI: 10.1007/s00216-018-1092-9 Miranda N. Emaus , Kevin D. Clark , Paige Hinners , Jared L. Anderson
Nucleic acid extraction and purification represents a major bottleneck in DNA analysis. Traditional methods for DNA purification often require reagents that may inhibit quantitative polymerase chain reaction (qPCR) if not sufficiently removed from the sample. Approaches that employ magnetic beads may exhibit lower extraction efficiencies due to sedimentation and aggregation. In this study, four hydrophobic magnetic ionic liquids (MILs) were investigated as DNA extraction solvents with the goal of improving DNA enrichment factors and compatibility with downstream bioanalytical techniques. By designing custom qPCR buffers, we directly incorporated DNA-enriched MILs including trihexyl(tetradecyl)phosphonium tris(hexafluoroacetylaceto)nickelate(II) ([P6,6,6,14+][Ni(hfacac)3−]), [P6,6,6,14+] tris(hexafluoroacetylaceto)colbaltate(II) ([Co(hfacac)3−]), [P6,6,6,14+] tris(hexafluoroacetylaceto)manganate(II) ([Mn(hfacac)3−]), or [P6,6,6,14+] tetrakis(hexafluoroacetylaceto)dysprosate(III) ([Dy(hfacac)4−]) into reaction systems, thereby circumventing the need for time-consuming DNA recovery steps. Incorporating MILs into the reaction buffer did not significantly impact the amplification efficiency of the reaction (91.1%). High enrichment factors were achieved using the [P6,6,6,14+][Ni(hfacac)3−] MIL for the extraction of single-stranded and double-stranded DNA with extraction times as short as 2 min. When compared to a commercial magnetic bead-based platform, the [P6,6,6,14+][Ni(hfacac)3−] MIL was capable of producing higher enrichment factors for single-stranded DNA and similar enrichment factors for double-stranded DNA. The MIL-based method was applied for the extraction and direct qPCR amplification of mutation prone-KRAS oncogene fragment in plasma samples.
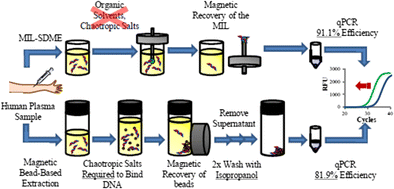
Magnetic ionic liquid solvents are shown to preconcentrate sufficient KRAS DNA template from an aqueous solution in as short as 2 min without using chaotropic salts or toxic organic solvents. By using custom-designed qPCR buffers, DNA can be directly amplified and quantified from four MILs examined in this study.
中文翻译:

使用与实时PCR兼容的磁性离子液体对DNA进行预浓缩以进行快速核酸定量
核酸提取和纯化代表了DNA分析的主要瓶颈。传统的DNA纯化方法通常需要试剂,如果不能从样品中充分去除,它们可能会抑制定量聚合酶链反应(qPCR)。由于沉积和聚集,采用磁珠的方法可能会表现出较低的提取效率。在这项研究中,以提高DNA富集因子和与下游生物分析技术的相容性为目标,研究了四种疏水性磁性离子液体(MIL)作为DNA提取溶剂。通过设计定制的qPCR缓冲剂,我们直接掺入DNA富集密耳包括三己基(十四烷基)鏻三(hexafluoroacetylaceto)镍(II)([P 6,6,6,14 + ] [镍(hfacac)3 -]),[P 6,6,6,14 + ]三(六氟乙酰乙酰)钴酸(II)([Co(hfacac)3 - ]),[P 6,6,6,14 + ]三(六氟乙酰乙酰)锰酸( II)([锰(hfacac)3 - ]),或[P 6,6,6,14 + ]四(hexafluoroacetylaceto)dysprosate(III)([镝(hfacac)4 - ])到反应系统,从而绕过需要耗时的DNA回收步骤。将MIL掺入反应缓冲液中不会显着影响反应的扩增效率(91.1%)。使用[P均达到高富集因子6,6,6,14 + ] [镍(hfacac)3 -] MIL用于提取单链和双链DNA,提取时间短至2分钟。当与商用基于磁珠的平台,[P 6,6,6,14 + ] [镍(hfacac)3 - ] MIL能够产生用于单链DNA以及类似的富集因子对于双更高富集因子的链DNA。基于MIL的方法用于血浆样品中易突变KRAS癌基因片段的提取和直接qPCR扩增。

已显示,磁性离子液体溶剂可在短短2分钟内从水溶液中预浓缩足够的KRAS DNA模板,而无需使用离液序列高的盐或有毒的有机溶剂。通过使用定制设计的qPCR缓冲液,可以从本研究中检测的四个MIL中直接扩增和定量DNA。











































 京公网安备 11010802027423号
京公网安备 11010802027423号