当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Piers’ borane-mediated hydrosilylation of epoxides and cyclic ethers†
Chemical Communications ( IF 4.3 ) Pub Date : 2018-06-07 00:00:00 , DOI: 10.1039/c8cc03741h Jianbo Zhang 1, 2, 2, 3, 4 , Sehoon Park 1, 2, 2, 3, 4 , Sukbok Chang 1, 2, 2, 3, 4
Chemical Communications ( IF 4.3 ) Pub Date : 2018-06-07 00:00:00 , DOI: 10.1039/c8cc03741h Jianbo Zhang 1, 2, 2, 3, 4 , Sehoon Park 1, 2, 2, 3, 4 , Sukbok Chang 1, 2, 2, 3, 4
Affiliation
|
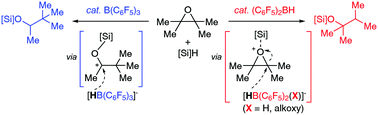
We report the first diarylborane-catalysed hydrosilylation of epoxides and cyclic ethers. Mechanistic studies on the in situ generated Piers’ borane (C6F5)2BH with hydrosilanes in the presence of an epoxide revealed that an alkyloxy(diaryl)borane (C6F5)2BOR is readily formed as a catalytically competent species for the outer-sphere hydrosilylation of epoxides and cyclic ethers.
中文翻译:

Piers硼烷介导的环氧化物和环醚的氢化硅烷化†
我们报道了环氧化物和环醚的第一个二芳基硼烷催化的氢化硅烷化反应。在环氧化物存在下与氢硅烷原位生成Piers's硼烷(C 6 F 5)2 BH的机理研究表明,烷氧基(二芳基)硼烷(C 6 F 5)2 BOR易于形成为催化活性物质用于环氧化物和环醚的外球氢化硅烷化。
更新日期:2018-06-07
中文翻译:

Piers硼烷介导的环氧化物和环醚的氢化硅烷化†
我们报道了环氧化物和环醚的第一个二芳基硼烷催化的氢化硅烷化反应。在环氧化物存在下与氢硅烷原位生成Piers's硼烷(C 6 F 5)2 BH的机理研究表明,烷氧基(二芳基)硼烷(C 6 F 5)2 BOR易于形成为催化活性物质用于环氧化物和环醚的外球氢化硅烷化。











































 京公网安备 11010802027423号
京公网安备 11010802027423号