当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Directed Remote Lateral Metalation: Highly Substituted 2‐Naphthols and BINOLs by In Situ Generation of a Directing Group
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2018-06-21 , DOI: 10.1002/anie.201805203 Jignesh J. Patel 1 , Marju Laars 1 , Wei Gan 1 , Johnathan Board 1 , Matthew O. Kitching 2 , Victor Snieckus 1
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2018-06-21 , DOI: 10.1002/anie.201805203 Jignesh J. Patel 1 , Marju Laars 1 , Wei Gan 1 , Johnathan Board 1 , Matthew O. Kitching 2 , Victor Snieckus 1
Affiliation
|
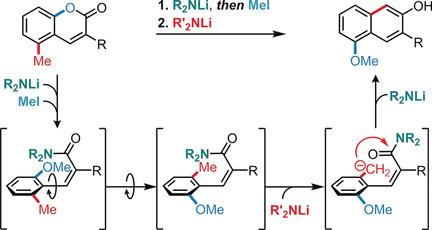
A general synthesis of highly substituted 2‐naphthols based on a new carbanionic reaction sequence is demonstrated. The reaction exploits the dual nature of lithium bases consisting of consecutive ring opening of readily available coumarins with either LiNEt2 or LiNiPr2 into Z‐cinnamamides, thus generating a directing group in situ and allowing, by conformational freedom, a lateral directed remote metalation for ring closure to give the aryl 2‐naphthols in good to excellent yields. These transformations can be combined to provide a more efficient one‐pot process. Mechanistic insight into the remote lateral metalation step, demonstrating the requirement of Z‐cinnamamide, is described. Application of this methodology to the synthesis of highly substituted 3,3′‐diaryl BINOL ligands is also reported.
中文翻译:

定向远程侧向金属化:定向基团的原位生成高度取代的2-萘酚和BINOL
演示了基于新的碳负离子反应序列的高度取代的2-萘酚的一般合成。该反应利用了锂碱的双重性质,即将容易获得的具有LiNEt 2或LiN i Pr 2的香豆素连续开环成Z肉桂酰胺,从而在原位生成一个导向基团,并通过构象自由度允许横向定向的远程金属化以进行闭环反应,得到芳基2-萘酚,收率好至极好。可以将这些转换组合起来,以提供更有效的单锅法。对远端横向金属化步骤的机械洞察力,证明了对Z的要求描述了肉桂酰胺。还报道了该方法在高度取代的3,3'-二芳基BINOL配体合成中的应用。
更新日期:2018-06-21
中文翻译:

定向远程侧向金属化:定向基团的原位生成高度取代的2-萘酚和BINOL
演示了基于新的碳负离子反应序列的高度取代的2-萘酚的一般合成。该反应利用了锂碱的双重性质,即将容易获得的具有LiNEt 2或LiN i Pr 2的香豆素连续开环成Z肉桂酰胺,从而在原位生成一个导向基团,并通过构象自由度允许横向定向的远程金属化以进行闭环反应,得到芳基2-萘酚,收率好至极好。可以将这些转换组合起来,以提供更有效的单锅法。对远端横向金属化步骤的机械洞察力,证明了对Z的要求描述了肉桂酰胺。还报道了该方法在高度取代的3,3'-二芳基BINOL配体合成中的应用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号